Product Images Actonel
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Actonel NDC 0430-0472 by Allergan, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
actonel 06
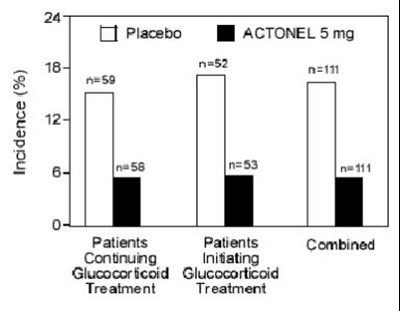
This appears to be a table displaying incidence percentages for patients continuing and initiating treatment with Actonel 5mg and placebo in conjunction with glucocorticoid treatment. The table shows 184 patients on Actonel 5mg continuing glucocorticoid treatment, with a 24% incidence rate. However, more context and information is needed to understand the full significance of these numbers.*
actonel 07
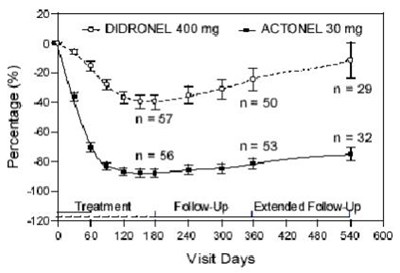
This text likely represents a table or chart showing the percentage of follow-up visits for patients taking DIDRONEL 400mg and ACTONEL 30mg over a period of time. However, without more information, it is difficult to interpret the specific data presented.*
actonel 08

Actonel 5 is a medication in the form of tablets containing alendronate sodium used as a treatment for bone diseases. The National Drug Code (NDC) for this medication is 0430-0471-15, and the package includes 30 tablets. It is recommended to dispense the enclosed medication guide to each patient before administration. This medication is only available with a prescription (Rx only).*
actonel 09

This text is a pharmaceutical label for Actonel, a medication used to treat osteoporosis. It contains the NDC code, dosage information, and an instruction to include a medication guide with the prescription. Because there is not enough context to determine the language of the label, it is unclear whether the "fip" in "Dispense the enclosed Medication Guide fip to each patient" is a meaningful word.*
actonel 0a

This appears to be a product label for Actonel tablets, which contain Ibandronate Sodium. The label includes information such as dosages (35mg), a National Drug Code (50229 00590) and a lot number (W34 138V1). Additionally, the label indicates that an enclosed patient information guide should be provided with each prescription.*
actonel 0c

This appears to be a medication guide for Actone, containing instructions on how to take the tablets, including dosage and timing. It advises patients to take one tablet in the morning with plain water, before eating or drinking anything else, and to avoid lying down or taking other medications for 30 minutes afterwards. The text also mentions an enclosed medication guide that should be dispensed to each patient. The last line includes the unrelated text "Apo xy".*
actonel 0e

This seems to be a package label for a 3 month pack of medication tablets called "Actonel" which contains 150mg of "risedronate sodium". The label includes information on how to dispense the tablets to patients along with a medication guide. There are also contact details for the manufacturer located at "201vg) AV 40 pemaes o5 0504".*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.