Product Images Xtandi
View Photos of Packaging, Labels & Appearance
- Structural formula of Enzalutamide - image 01
- Figure 2. Effect of XTANDI on Other Drugs - image 02
- Figure 1. Effects of Other Drugs and Intrinsic/Extrinsic Factors on XTANDI - image 03
- Figure 3. Kaplan-Meier Curves of Overall Survival in AFFIRM - image 04
- Figure 4. Kaplan-Meier Curves of Overall Survival in PREVAIL - image 05
- Figure 5. Kaplan-Meier Curves of Radiographic Progression-free Survival in PREVAIL - image 06
- Figure 6. Kaplan-Meier Curves of Radiographic Progression-free Survival in TERRAIN - image 07
- Figure 7. Kaplan-Meier Curves of Metastasis-free Survival in PROSPER - image 08
- Xtandi (enzalutamide) capsules 40 mg label - image 10
- Figure 9. Kaplan-Meier Curves of rPFS in ARCHES (Intent-to-Treat Analysis) - image 12
- Figure 8. Kaplan-Meier Curves of Overall Survival in PROSPER - image 13
- image 14
- image 15
Product Label Images
The following 13 images provide visual information about the product associated with Xtandi NDC 0469-0125 by Astellas Pharma Us, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Structural formula of Enzalutamide - image 01
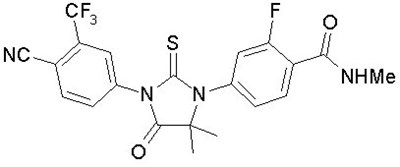
The text "NC NHMe" likely refers to two chemical compounds: N-cyanomethylamine and N-methylformamide, which may have applications in organic synthesis and pharmaceuticals.*
Figure 2. Effect of XTANDI on Other Drugs - image 02

This text provides information about a pharmaceutical product called Xtandi, which contains enzalutamide as an active ingredient. The recommended dosage is not specified but the product comes in tablet form with a strength of 40 mg. The product is manufactured by PharmaCeuticals LLC and marketed by Al Pharms US. The National Drug Code (NDC) for this product is 0463.0625-98 and it is only available with a prescription (Rx Only).*
Figure 1. Effects of Other Drugs and Intrinsic/Extrinsic Factors on XTANDI - image 03

Recommended dosage for the medication Xtandi is indicated by NDC 0469-0725-60. The medication should be stored at a temperature between 4-2°C. Excursions permitted only up to 15°C. The medication is manufactured by Phamaceubials LLC for Astetas Phara US, Inc. The product comes in a pack of 60 tablets and should be kept out of reach of children.*
Figure 3. Kaplan-Meier Curves of Overall Survival in AFFIRM - image 04

This text appears to present data from a clinical trial comparing the survival of patients treated with Xtandi against a placebo. The data is presented graphically and shows that the survival rate of patients treated with Xtandi was higher compared to those who received placebo. The median survival for Xtandi was 184 months whereas it was only 13.6 months for placebo. The hazard ratio for Xtandi was 0.63, which further supports its efficacy. There is information about the sample size and confidence interval as well. Overall, this text seems to be describing the positive impact of Xtandi on patient survival in a clinical trial context.*
Figure 4. Kaplan-Meier Curves of Overall Survival in PREVAIL - image 05
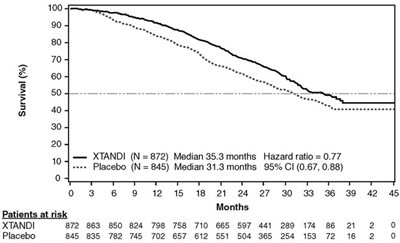
This is a table showing the survival rates of patients who were given XTANDI and Placebo treatments. Median survival for patients who took XTANDI was 35.3 months, compared to 31.3 months for those who took Placebo. The hazard ratio was 0.77, and the 95% Cl range was 0.67 to 0.88. The table also provides a graph showing the percentage survival rate over time for both groups.*
Figure 5. Kaplan-Meier Curves of Radiographic Progression-free Survival in PREVAIL - image 06

The text describes the Radiographic Progression-Free Survival (%) of two drugs - XTANDI and Placebo - used for treatment. The table shows that XTANDI has a median not available (NR) of progression-free survival with a hazard ratio of 0.17, while placebo has a median of 3.7 months with a 95% confidence interval of 0.14-0.21 and a p-value of less than 0.0001. The data is tracked for 21 months and shows the number of patients surviving at different stages.*
Figure 6. Kaplan-Meier Curves of Radiographic Progression-free Survival in TERRAIN - image 07

This is a comparison of radiographic progression-free survival rates between XTANDI and Bicalutamide treatments in patients with a certain medical condition. The study had a sample size of 184 patients for XTANDI treatment and 191 patients for Bicalutamide treatment. The median progression-free survival for patients receiving XTANDI was 19.5 months, with a hazard ratio of 0.60. The median progression-free survival for patients receiving Bicalutamide was 13.4 months, with a 95% confidence interval between 0.43 and 0.83. The graph shows the percentage of patients with radiographic progression-free survival over time, with XTANDI showing better progression-free survival rates compared to Bicalutamide. The study lasted for up to 27 months.*
Figure 7. Kaplan-Meier Curves of Metastasis-free Survival in PROSPER - image 08

The text seems to show data of a clinical study comparing the survival rates between two treatment groups for metastasis. The treatment with XTANDI had a median metastasis-free survival of 36.6 months, while the placebo group had a median survival of 14.7 months. The hazard ratio of the placebo group was 0.29. The study had 933 participants. The table includes a visual representation of the survival curves and the number of patients at risk in each treatment group over time in months.*
Xtandi (enzalutamide) capsules 40 mg label - image 10

This text provides information about dosage and storage of medication in the form of capsules. The dosage instructions are not given explicitly and the users are advised to refer to the package insert. The medication should be stored in a dry place, and the container must be kept tightly closed. The temperature for storage should be between 68°F to 77°F, with excursions permitted from 59°F to 86°F. The medication is available in a packet containing 120 capsules, and the manufacturer is Catalent Pharma Solutions LLC for Astellas Pharma US Inc. The users are advised to keep the medication away from children.*
Figure 9. Kaplan-Meier Curves of rPFS in ARCHES (Intent-to-Treat Analysis) - image 12

Not available.*
image 15

XTANDI (enzalutamide) is a medication that comes in tablet form. The recommended dosage and storage instructions are provided on the label, including the warning not to cut, crush, or chew the tablets. The medication should be stored within a specific temperature range to maintain its strength. This drug is manufactured by Pfizer Pharmaceuticals and Astellas Pharma US, Inc. Children should not have access to the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

