Product Images Prograf
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 28 images provide visual information about the product associated with Prograf NDC 0469-0657 by Astellas Pharma Us, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image 08
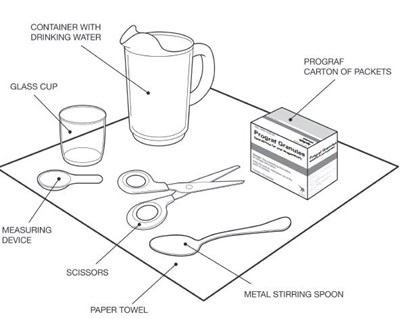
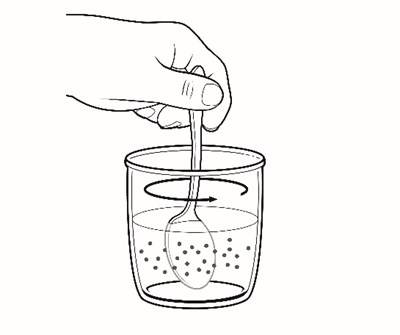
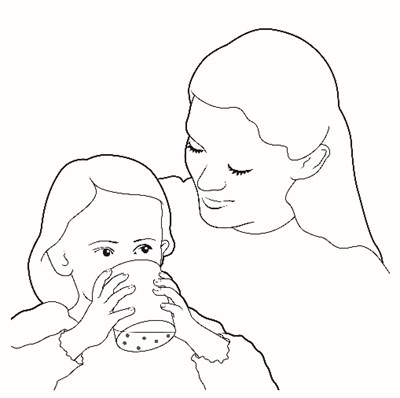
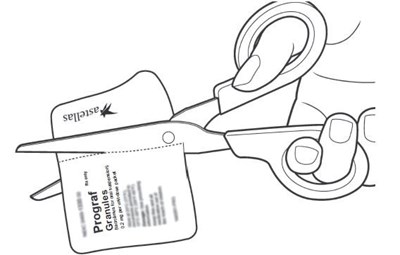
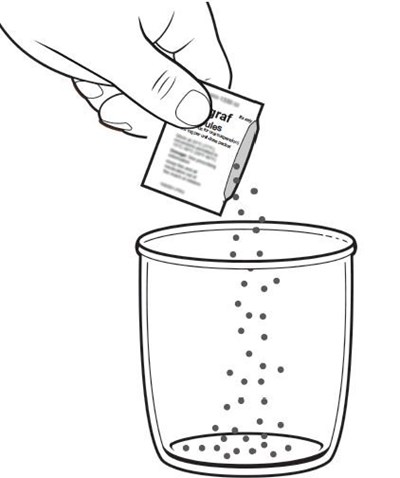
This text is a list of items that includes a container of drinking water, a carton of packets of a substance named Prograf, a glass cup, a measuring device, scissors, a metal stirring spoon, and a paper towel. There is no further information available in this text.*
image 15

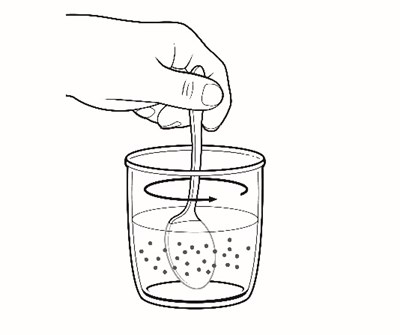
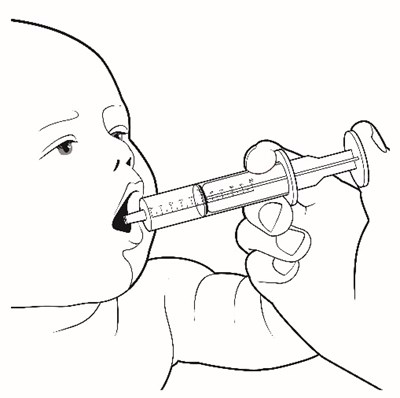
This appears to be a list of various items included in a container. The contents include drinking water, a plunger, oral medication in a carton of packets, a syringe, a glass cup, a measuring device, scissors, a metal stirring spoon, and a paper towel. It is unclear what the container is intended for.*
image 23

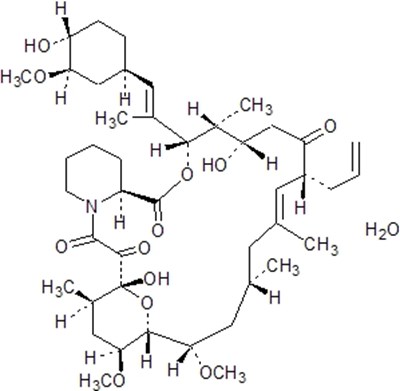
Prograf (tacrolimus) capsules are a prescription medication that come in a pack of 100 capsules, produced in Japan, by Astellas Ireland Co., Ltd. It is marketed by Astellas Pharma US, Inc. Prograf capsules contain 0.5mg of Tacrolimus along with lactose, hydroxypropyl methylcellulose 2910, croscarmellose sodium, and magnesium stearate inside them. The capsules should be stored at a temperature of 25°C (77°F) with excursions allowed between 15°C-30°C (59°F-86°F). For more detailed dosage information please see the package insert.*
image 24

Prograf capsules contain Tacrolimus and are used to suppress the immune system after organ transplant surgery. These capsules are manufactured by Astellas Ireland Co., Ltd. in Japan and marketed by Astellas Pharma US, Inc. in Northbrook, Illinois. Each hard gelatin capsule contains 1 mg of Tacrolimus, along with lactose, hydroxypropyl methylcellulose 2910, croscarmellose sodium and magnesium stearate as inactive ingredients. The recommended storage temperature for the capsules is 25°C (77°F), with excursions allowed between 15°C-30°C (59°F-86°F). It is advised to refer to the package insert for dosage information.*
image 25
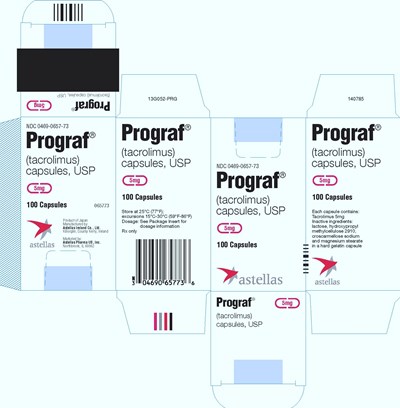
Prograf (tacrolimus) capsules are a medication with the active ingredient Tacrolimus. It comes in a pack of 100 capsules by Astellas Ireland Co., Ltd. and sold by Astellas Pharma US, Inc. The recommended storage is between 15°C to 30°C. It is a prescription drug that must be taken as per dosage information provided in the package insert. The capsules are made of lactose, hydroxypropyl methylcellulose 2910, croscarmellose sodium, and magnesium stearate in a hard gelatin capsule.*
image 26

Prograf® (tacrolimus) is a sterile injectable solution for intravenous use only after dilution. It is available in 10 (1 mL) ampules containing 5 mg of Tacrolimus, a drug used to prevent organ transplant rejection. The solution also contains Polyoxyl 60 Hydrogenated Castor Oil and Dehydrated Alcohol. Dosage information can be found in the package insert. The solution should be stored between 5°C and 25°C (41°F- 77°F). Prograf® is a registered trademark of Astellas Pharma Inc.*
image 27

This is a label for the drug Prograf® Granules, containing tacrolimus for oral suspension. The label provides information for mixing and preparing the medication to be taken. The dosage information is available on the prescribing information. The medication should be kept away from children. The label also includes a 2D barcode and variable code area, and it contains 50 unit-dose packets.*
image 28

This is a label of a medication called Prograf®Granules, containing Tacrolimus, for oral suspension. The label provides instructions on how to mix and prepare the granules, dosage information, and a warning to keep it out of the reach of children. The label also includes a variable code area and a 2D barcode.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.