Product Images Myrbetriq
View Photos of Packaging, Labels & Appearance
- Mirabegron structural formula - image 01
- Astellas logo - image 02
- Astellas logo - image 03
- logo.jpg - image 04
- logo.jpg - image 05
- 25mg Extended release tablet carton - image 06
- 50mg Extended release tablet carton - image 07
- Figure 1 - image 08
- Figure 2 - image 09
- Figure 3 - image 10
- Figure 4 - image 11
- Figure 5 - image 12
- Figure 6 - image 13
- Figure 7 - image 14
- Figure 8 - image 15
- Figure 9 - image 16
- Figure 10 - image 17
- Myrbetriq Granules carton image - image 18
Product Label Images
The following 18 images provide visual information about the product associated with Myrbetriq NDC 0469-5020 by Astellas Pharma Us, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
25mg Extended release tablet carton - image 06

Myrbetriq® (mirabegron extended-release tablets) is a medication taken once daily that comes in 25 mg tablets. The tablets should be swallowed whole and should not be crushed or chewed. This prescription medication is prescribed in 30-tablet quantities. All prescription medications, including Myrbetriq, should be kept out of reach of children.*
Figure 1 - image 08
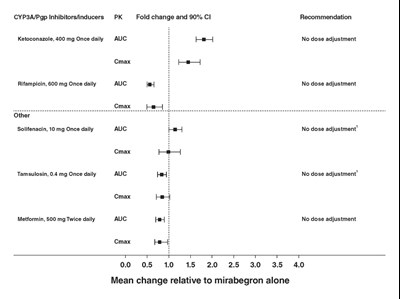
The text describes a chart that shows the effect of inhibitors/inducers of CYP3A/Pgp on PK fold change and 90% confidence interval. The chart includes recommended dose adjustments for ketoconazole and rifampicin, with no changes needed for these medications. The chart also includes solifenacin, tamsulosin, and metformin, with no dose adjustments recommended for these medications either. There is a graph as well, displaying the mean change relative to mirabegron alone over time.*
Figure 2 - image 09

This text provides information on the recommended dose adjustments for various drugs that are metabolized by the enzymes CYP3A4 and CYP2D6. Solifenacin, ethinylestradiol, and levonorgestrel do not require any dose adjustment. Desipramine and metoprolol should be used with caution, while tamsulosin and metformin do not require any dose adjustment. Digoxin requires a starting dose adjustment, and for warfarin, the dosing should be guided by footnote 3. Information about the mean change relative to the substrate alone is also provided.*
Figure 7 - image 14

This appears to be a table showing mean changes from baseline for different treatment groups in a clinical trial related to a drug called "mirabegron" at different time points. The drug was tested at two different doses: 25 mg and 50 mg. The table shows data for three groups: a placebo group (n=262), a group receiving mirabegron 25mg (n=254), and a group receiving mirabegron 50mg (n=257). However, without more context it is difficult to interpret the table further.*
Figure 8 - image 15

This appears to be a table or graph displaying numerical data related to a study or clinical trial. It shows the mean change from baseline for participants receiving either a placebo or two doses of a drug called Mirabegron at 4, 8, and 12 weeks. However, without further context or explanation it is difficult to interpret the data or its significance.*
Figure 10 - image 17

This text appears to be a chart depicting the adjusted change from baseline in some sort of study or experiment. The chart includes various measurements taken at different time periods (baseline, week 4, week 8, and week 12), and different dosages (25mg and 50mg). It's difficult to determine what exactly is being measured without more context, but it appears to be some sort of percent change in response to different treatments.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.











