Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
- Carton Stock No.: 5601 (Principal Panel) - Carton 5601A
- Carton Stock No.: 5601 - Carton 5601B
- Carton Stock No.: 5602 (Principal Display) - Carton 5602A
- Carton Stock No.: 5602 - Carton 5602B
- Carton Stock No.: 5603 (Principal Panel) - Carton 5603A
- Carton Stock No.: 5603 - Carton 5603B
- Carton Stock No.: 5604 (Principal Panel) - Carton 5604A
- Carton Stock No.: 5604 - Carton 5604B
- Carton Stock No.: 5605 (Principal Panel) - Carton 5605A
- Carton Stock No.: 5605 - Carton 5605B
- Carton Stock No.: 5606 (Principal Panel) - Carton 5606A
- Carton Stock No.: 5606 - Carton 5606B
- Carton Stock No.: 5607 (Principal Panel) - Carton 5607A
- Carton Stock No.: 5607 - Carton 5607B
- Carton Stock No.: 5608 (Principal Panel) - Carton 5608
- Instructions 1 - enoxaparin 00
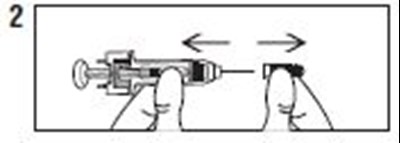
- Instructions 2 - enoxaparin 01
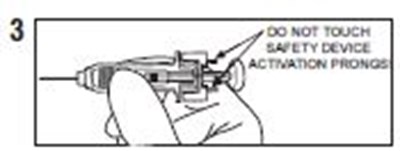
- Instructions 3 - enoxaparin 02
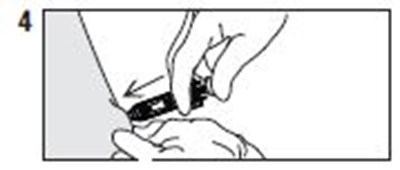
- Instructions 4 - enoxaparin 03
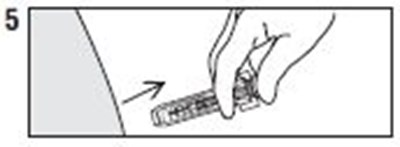
- Instructions 5 - enoxaparin 04
- Instructions 6 - enoxaparin 05
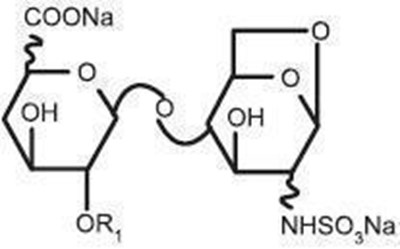
- Chemical Structure - enoxaparin 06
- Chemical Structure - enoxaparin 07
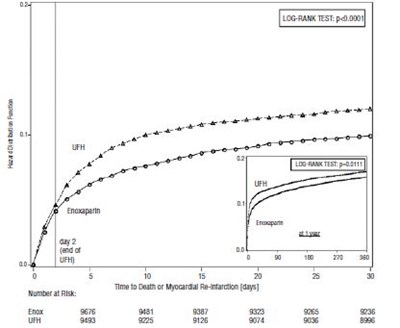
- Figure 1 - enoxaparin 08
- Figure 2 - enoxaparin 09
Product Label Images
The following 25 images provide visual information about the product associated with Enoxaparin Sodium NDC 0548-5606 by Amphastar Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carton Stock No.: 5601 (Principal Panel) - Carton 5601A

This is a description of Enoxaparin Sodium Injection, a medication used for subcutaneous injection. It comes in a single dose syringe with 30 mg/0.3 mL strength and has automated safety devices. The brand names mentioned are AxoNLY and RxonLY.*
Carton Stock No.: 5601 - Carton 5601B

This is a package of Enoxaparin sodium injection containing 10 single-dose syringes with a strength of 30 mg. It is meant for subcutaneous injection and comes with instructions.*
Carton Stock No.: 5602 (Principal Display) - Carton 5602A

This is a description of Enoxaparin sodium injection in a single dose syringe with an automatic safety device. The injection contains 40 mg/0.4 mL of Enoxaparin sodium and is intended for subcutaneous injection.*
Carton Stock No.: 5603 (Principal Panel) - Carton 5603A

This is a product description for Enoxaparin Sodium Injection, which is available in a dosage strength of 60 mg/0.6 mL. It is indicated for subcutaneous injection and is available in both single-dose syringes and multi-dose vials. The remaining text is not readable.*
Carton Stock No.: 5603 - Carton 5603B

This is a description of a medication called enoxaparin sodium injection, which comes in single-dose syringes of 60mg. The price of this product is £261.05 and it is used for subcutaneous injection. The text includes dosage instructions and warnings to keep it out of reach of children.*
Carton Stock No.: 5604 (Principal Panel) - Carton 5604A

This is a package of Enoxaparin Sodium Injection containing 80 mg/0.8 mL in 50 single-dose syringes with automated safety device for subcutaneous injection. The text is partially cut off, and there are some errors, but the product name, dosage, administration, and safety information are discernible.*
Carton Stock No.: 5605 - Carton 5605B

This is a description of a medication called Enoxaparin Sodium Injection, which comes in a package of 10 single-dose syringes that each contain 100 mg of the medication. It is meant for subcutaneous injection. The text also includes some numbers and a warning about storage temperature. There is some garbled text at the beginning that is not readable or available.*
Carton Stock No.: 5606 (Principal Panel) - Carton 5606A

ENOXAPARIN SODIUM INJECTION is a medication administered through subcutaneous injection using single dose syringes with an automated safety device. Each syringe contains 120 mg of medication in 0.8 mL solution. This information is intended for medical professionals only.*
Carton Stock No.: 5606 - Carton 5606B

This is a description of Enoxaparin Sodium Injection, which comes in 10 single dose syringes of 120mg each. Enoxaparin Sodium is to be injected subcutaneously and is used for the prevention of blood clots. The text also includes some warnings and precautions for the proper use of the medication.*
Carton Stock No.: 5607 (Principal Panel) - Carton 5607A

ENOXAPARIN SODIUM INJECTION is a prescription medication available in single-dose syringes with automatic safety devices, containing 150mg/mL of the active ingredient. It is intended for use as a subcutaneous injection.*
Figure 1 - enoxaparin 08

The given text includes a table that possibly presents a study related to risk reduction due to some medication or treatment for patients with different medical conditions. The presented variables include patient's sex, age, infarction location, diabetes, prior Mi, fibrinolytic agents, time to treatment, and other parameters. The relative risk reduction for each subgroup is given in percentages. The study compares two different types of fibrinolytic agents and shows that UFH is a better option for risk reduction.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.