Product Images Valproic
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Valproic NDC 0591-4012 by Actavis Pharma, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The following structural formula for Valproic acid (pKa 4.8) has a molecular weight of 144 and occurs as a colorless liquid with a characteristic odor. It is slightly soluble in water (1.3 mg/mL) and very soluble in organic solvents. - image 1
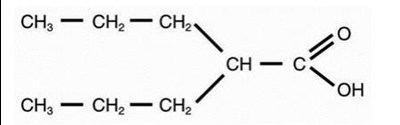
image - image 4

This is a description for Valproic Acid Capsules, USP with NDC 0591-4012-01. Each capsule contains 250mg of Valproic Acid, USP. The medication guide provided separately should be dispensed to each patient by a pharmacist. It is for Rx only with 100 capsules. The capsules should be stored in a tight, light-resistant container at 15° to 30°C (59° to 86°F) and protected from humidity, light, and moisture. The full prescribing information is in the package insert. It is manufactured by Catalent Pharma Solutions, LLC, St. Petersburg, FL 33716-1016, and manufactured for Teva Pharmaceuticals USA, Inc., Parsippany, N) 07054. The description was last revised in 05/2021.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

