FDA Label for Synjardy
View Indications, Usage & Precautions
- WARNING: LACTIC ACIDOSIS
- 1 INDICATIONS AND USAGE
- OTHER
- 2.1 PRIOR TO INITIATION OF SYNJARDY
- 2.2 RECOMMENDED DOSAGE AND ADMINISTRATION
- 2.3 DOSAGE RECOMMENDATIONS IN PATIENTS WITH RENAL IMPAIRMENT
- 2.4 DISCONTINUATION FOR IODINATED CONTRAST IMAGING PROCEDURES
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 LACTIC ACIDOSIS
- 5.2 KETOACIDOSIS
- 5.3 VOLUME DEPLETION
- 5.4 UROSEPSIS AND PYELONEPHRITIS
- 5.5 HYPOGLYCEMIA WITH CONCOMITANT USE WITH INSULIN AND INSULIN SECRETAGOGUES
- 5.6 NECROTIZING FASCIITIS OF THE PERINEUM (FOURNIER'S GANGRENE)
- 5.7 GENITAL MYCOTIC INFECTIONS
- 5.8 HYPERSENSITIVITY REACTIONS
- 5.9 VITAMIN B12 DEFICIENCY
- 6 ADVERSE REACTIONS
- 6.1 CLINICAL TRIALS EXPERIENCE
- 6.2 POSTMARKETING EXPERIENCE
- 7 DRUG INTERACTIONS
- 8.3 FEMALES AND MALES OF REPRODUCTIVE POTENTIAL
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 8.6 RENAL IMPAIRMENT
- 8.7 HEPATIC IMPAIRMENT
- 10 OVERDOSAGE
- 11 DESCRIPTION
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- SPL MEDGUIDE
- PRINCIPAL DISPLAY PANEL - 5 MG/500 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 5 MG/1000 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 12.5 MG/500 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 12.5 MG/1000 MG TABLET BOTTLE LABEL
Synjardy Product Label
The following document was submitted to the FDA by the labeler of this product Boehringer Ingelheim Pharmaceuticals, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Warning: Lactic Acidosis
Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1)].
Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.1), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7)].
If metformin-associated lactic acidosis is suspected, immediately discontinue SYNJARDY and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1)].
1 Indications And Usage
SYNJARDY is a combination of empagliflozin and metformin hydrochloride (HCl) indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Empagliflozin is indicated to reduce the risk of cardiovascular death in adults with type 2 diabetes mellitus and established cardiovascular disease [see Clinical Studies (14)].
Other
Limitation of Use
SYNJARDY is not recommended in patients with type 1 diabetes mellitus. It may increase the risk of diabetic ketoacidosis in these patients [see Warnings and Precautions (5.2)].
Renal Impairment: The postmarketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient's renal function include [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
- Before initiating SYNJARDY, obtain an estimated glomerular filtration rate (eGFR).
- SYNJARDY is contraindicated in patients with an eGFR below 30 mL/min/1.73 m2 [see Contraindications (4)].
- Obtain an eGFR at least annually in all patients taking SYNJARDY. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
- Ketoacidosis
- Urosepsis and Pyelonephritis
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene)
- Angioedema
- Acute Kidney Injury
- Skin Reactions (e.g., rash, urticaria)
- Cholestatic, hepatocellular, and mixed hepatocellular liver injury
Drug Interactions: The concomitant use of SYNJARDY with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance or increase metformin accumulation [see Drug Interactions (7)]. Therefore, consider more frequent monitoring of patients.
Age 65 or Greater: The risk of metformin-associated lactic acidosis increases with the patient's age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients [see Use in Specific Populations (8.5)].
Radiological Studies with Contrast: Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop SYNJARDY at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR less than 60 mL/min/1.73 m2; in patients with a history of hepatic impairment, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart SYNJARDY if renal function is stable.
Surgery and Other Procedures: Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension and renal impairment. SYNJARDY should be temporarily discontinued while patients have restricted food and fluid intake.
Hypoxic States: Several of the postmarketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur, discontinue SYNJARDY.
Excessive Alcohol Intake: Alcohol potentiates the effect of metformin on lactate metabolism and this may increase the risk of metformin-associated lactic acidosis. Warn patients against excessive alcohol intake while receiving SYNJARDY.
Hepatic Impairment: Patients with hepatic impairment have developed cases of metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of SYNJARDY in patients with clinical or laboratory evidence of hepatic disease.
Empagliflozin Add-On Combination Therapy with Metformin
In a 24-week placebo-controlled trial of empagliflozin 10 mg and 25 mg administered once daily added to metformin, there were no adverse reactions reported regardless of investigator assessment of causality in ≥5% of patients and more commonly than in patients given placebo.
Empagliflozin Add-On Combination Therapy with Metformin and Sulfonylurea
In a 24-week placebo-controlled trial of empagliflozin 10 mg and 25 mg administered once daily added to metformin and sulfonylurea, adverse reactions reported regardless of investigator assessment of causality in ≥5% of patients and more commonly than in patients given placebo are presented in Table 1 (see also Table 4).
| Adverse Reactions | Placebo (%) n=225 | Empagliflozin 10 mg (%) n=224 | Empagliflozin 25 mg (%) n=217 |
|---|---|---|---|
| Hypoglycemia | 9.8 | 15.6 | 12.9 |
| Urinary tract infection | 6.7 | 9.4 | 6.9 |
| Nasopharyngitis | 4.9 | 8.0 | 6.0 |
Empagliflozin
The data in Table 2 are derived from a pool of four 24-week placebo-controlled trials and 18-week data from a placebo-controlled trial with basal insulin. Empagliflozin was used as monotherapy in one trial and as add-on therapy in four trials [see Clinical Studies (14)].
These data reflect exposure of 1976 patients to empagliflozin with a mean exposure duration of approximately 23 weeks. Patients received placebo (N=995), empagliflozin 10 mg (N=999), or empagliflozin 25 mg (N=977) once daily. The mean age of the population was 56 years and 3% were older than 75 years of age. More than half (55%) of the population was male; 46% were White, 50% were Asian, and 3% were Black or African American. At baseline, 57% of the population had diabetes more than 5 years and had a mean hemoglobin A1c (HbA1c) of 8%. Established microvascular complications of diabetes at baseline included diabetic nephropathy (7%), retinopathy (8%), or neuropathy (16%). Baseline renal function was normal or mildly impaired in 91% of patients and moderately impaired in 9% of patients (mean eGFR 86.8 mL/min/1.73 m2).
Table 2 shows common adverse reactions (excluding hypoglycemia) associated with the use of empagliflozin. The adverse reactions were not present at baseline, occurred more commonly on empagliflozin than on placebo and occurred in greater than or equal to 2% of patients treated with empagliflozin 10 mg or empagliflozin 25 mg.
| Adverse Reactions | Placebo (%) N=995 | Empagliflozin 10 mg (%) N=999 | Empagliflozin 25 mg (%) N=977 |
|---|---|---|---|
| aPredefined adverse event grouping, including, but not limited to, urinary tract infection, asymptomatic bacteriuria, cystitis | |||
| bFemale genital mycotic infections include the following adverse reactions: vulvovaginal mycotic infection, vaginal infection, vulvitis, vulvovaginal candidiasis, genital infection, genital candidiasis, genital infection fungal, genitourinary tract infection, vulvovaginitis, cervicitis, urogenital infection fungal, vaginitis bacterial. Percentages calculated with the number of female subjects in each group as denominator: placebo (N=481), empagliflozin 10 mg (N=443), empagliflozin 25 mg (N=420). | |||
| cPredefined adverse event grouping, including, but not limited to, polyuria, pollakiuria, and nocturia | |||
| dMale genital mycotic infections include the following adverse reactions: balanoposthitis, balanitis, genital infections fungal, genitourinary tract infection, balanitis candida, scrotal abscess, penile infection. Percentages calculated with the number of male subjects in each group as denominator: placebo (N=514), empagliflozin 10 mg (N=556), empagliflozin 25 mg (N=557). | |||
| Urinary tract infectiona | 7.6 | 9.3 | 7.6 |
| Female genital mycotic infectionsb | 1.5 | 5.4 | 6.4 |
| Upper respiratory tract infection | 3.8 | 3.1 | 4.0 |
| Increased urinationc | 1.0 | 3.4 | 3.2 |
| Dyslipidemia | 3.4 | 3.9 | 2.9 |
| Arthralgia | 2.2 | 2.4 | 2.3 |
| Male genital mycotic infectionsd | 0.4 | 3.1 | 1.6 |
| Nausea | 1.4 | 2.3 | 1.1 |
Thirst (including polydipsia) was reported in 0%, 1.7%, and 1.5% for placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively.
Volume Depletion
Empagliflozin causes an osmotic diuresis, which may lead to intravascular volume contraction and adverse reactions related to volume depletion. In the pool of five placebo-controlled clinical trials, adverse reactions related to volume depletion (e.g., blood pressure (ambulatory) decreased, blood pressure systolic decreased, dehydration, hypotension, hypovolemia, orthostatic hypotension, and syncope) were reported by 0.3%, 0.5%, and 0.3% of patients treated with placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively. Empagliflozin may increase the risk of hypotension in patients at risk for volume contraction [see Use in Specific Populations (8.5, 8.6)].
Increased Urination
In the pool of five placebo-controlled clinical trials, adverse reactions of increased urination (e.g., polyuria, pollakiuria, and nocturia) occurred more frequently on empagliflozin than on placebo (see Table 2). Specifically, nocturia was reported by 0.4%, 0.3%, and 0.8% of patients treated with placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively.
Hypoglycemia
The incidence of hypoglycemia by study is shown in Table 3. The incidence of hypoglycemia increased when empagliflozin was administered with insulin or sulfonylurea.
| aOverall hypoglycemic events: plasma or capillary glucose of less than or equal to 70 mg/dL | |||
| bSevere hypoglycemic events: requiring assistance regardless of blood glucose | |||
| cTreated set (patients who had received at least one dose of study drug) | |||
| dInsulin dose could not be adjusted during the initial 18 week treatment period | |||
| Monotherapy (24 weeks) | Placebo (n=229) | Empagliflozin 10 mg (n=224) | Empagliflozin 25 mg (n=223) |
| Overall (%) | 0.4 | 0.4 | 0.4 |
| Severe (%) | 0 | 0 | 0 |
| In Combination with Metformin (24 weeks) | Placebo + Metformin (n=206) | Empagliflozin 10 mg + Metformin (n=217) | Empagliflozin 25 mg + Metformin (n=214) |
| Overall (%) | 0.5 | 1.8 | 1.4 |
| Severe (%) | 0 | 0 | 0 |
| In Combination with Metformin + Sulfonylurea (24 weeks) | Placebo (n=225) | Empagliflozin 10 mg + Metformin + Sulfonylurea (n=224) | Empagliflozin 25 mg + Metformin + Sulfonylurea (n=217) |
| Overall (%) | 8.4 | 16.1 | 11.5 |
| Severe (%) | 0 | 0 | 0 |
| In Combination with Pioglitazone +/- Metformin (24 weeks) | Placebo (n=165) | Empagliflozin 10 mg + Pioglitazone +/- Metformin (n=165) | Empagliflozin 25 mg + Pioglitazone +/- Metformin (n=168) |
| Overall (%) | 1.8 | 1.2 | 2.4 |
| Severe (%) | 0 | 0 | 0 |
| In Combination with Basal Insulin +/-Metformin (18 weeksd) | Placebo (n=170) | Empagliflozin 10 mg (n=169) | Empagliflozin 25 mg (n=155) |
| Overall (%) | 20.6 | 19.5 | 28.4 |
| Severe (%) | 0 | 0 | 1.3 |
| In Combination with MDI Insulin +/-Metformin (18 weeksd) | Placebo (n=188) | Empagliflozin 10 mg (n=186) | Empagliflozin 25 mg (n=189) |
| Overall (%) | 37.2 | 39.8 | 41.3 |
| Severe (%) | 0.5 | 0.5 | 0.5 |
Genital Mycotic Infections
In the pool of five placebo-controlled clinical trials, the incidence of genital mycotic infections (e.g., vaginal mycotic infection, vaginal infection, genital infection fungal, vulvovaginal candidiasis, and vulvitis) was increased in patients treated with empagliflozin compared to placebo, occurring in 0.9%, 4.1%, and 3.7% of patients randomized to placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively. Discontinuation from study due to genital infection occurred in 0% of placebo-treated patients and 0.2% of patients treated with either empagliflozin 10 or 25 mg.
Genital mycotic infections occurred more frequently in female than male patients (see Table 2).
Phimosis occurred more frequently in male patients treated with empagliflozin 10 mg (less than 0.1%) and empagliflozin 25 mg (0.1%) than placebo (0%).
Urinary Tract Infections
In the pool of five placebo-controlled clinical trials, the incidence of urinary tract infections (e.g., urinary tract infection, asymptomatic bacteriuria, and cystitis) was increased in patients treated with empagliflozin compared to placebo (see Table 2). Patients with a history of chronic or recurrent urinary tract infections were more likely to experience a urinary tract infection. The rate of treatment discontinuation due to urinary tract infections was 0.1%, 0.2%, and 0.1% for placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively.
Urinary tract infections occurred more frequently in female patients. The incidence of urinary tract infections in female patients randomized to placebo, empagliflozin 10 mg, and empagliflozin 25 mg was 16.6%, 18.4%, and 17.0%, respectively. The incidence of urinary tract infections in male patients randomized to placebo, empagliflozin 10 mg, and empagliflozin 25 mg was 3.2%, 3.6%, and 4.1%, respectively [see Use in Specific Populations (8.5)].
Metformin
The most common (>5%) established adverse reactions due to initiation of metformin therapy are diarrhea, nausea/vomiting, flatulence, abdominal discomfort, indigestion, asthenia, and headache.
Laboratory Tests
Empagliflozin
Increases in Serum Creatinine and Decreases in eGFR: Initiation of empagliflozin causes an increase in serum creatinine and decrease in eGFR within weeks of starting therapy and then these changes stabilize. In a study of patients with moderate renal impairment, larger mean changes were observed. In a long-term cardiovascular outcomes trial, the increase in serum creatinine and decrease in eGFR generally did not exceed 0.1 mg/dL and -9.0 mL/min/1.73 m2, respectively, at Week 4, and reversed after treatment discontinuation, suggesting acute hemodynamic changes may play a role in the renal function changes observed with empagliflozin.
Increase in Low-Density Lipoprotein Cholesterol (LDL-C): Dose-related increases in low-density lipoprotein cholesterol (LDL-C) were observed in patients treated with empagliflozin. LDL-C increased by 2.3%, 4.6%, and 6.5% in patients treated with placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively. The range of mean baseline LDL-C levels was 90.3 to 90.6 mg/dL across treatment groups.
Increase in Hematocrit: In a pool of four placebo-controlled studies, median hematocrit decreased by 1.3% in placebo and increased by 2.8% in empagliflozin 10 mg and 2.8% in empagliflozin 25 mg-treated patients. At the end of treatment, 0.6%, 2.7%, and 3.5% of patients with hematocrits initially within the reference range had values above the upper limit of the reference range with placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively.
Metformin
Decrease in Vitamin B12: In metformin clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients.
Empagliflozin
Metformin hydrochloride
Risk Summary
Based on animal data showing adverse renal effects from empagliflozin, SYNJARDY is not recommended during the second and third trimesters of pregnancy.
The limited available data with SYNJARDY or empagliflozin in pregnant women are not sufficient to determine a drug-associated risk for major birth defects and miscarriage. Published studies with metformin use during pregnancy have not reported a clear association with metformin and major birth defect or miscarriage risk (see Data). There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy (see Clinical Considerations).
In animal studies, empagliflozin, a component of SYNJARDY, resulted in adverse renal changes in rats when administered during a period of renal development corresponding to the late second and third trimesters of human pregnancy. Doses approximately 13-times the maximum clinical dose caused renal pelvic and tubule dilatations that were reversible. No adverse developmental effects were observed when metformin was administered to pregnant rats or rabbits (see Data).
The estimated background risk of major birth defects is 6% to 10% in women with pre-gestational diabetes with a HbA1c >7 and has been reported to be as high as 20% to 25% in women with HbA1c >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk: Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data
Human Data
Published data from postmarketing studies have not reported a clear association with metformin and major birth defects, miscarriage, or adverse maternal or fetal outcomes when metformin was used during pregnancy. However, these studies cannot definitely establish the absence of any metformin-associated risk because of methodological limitations, including small sample size and inconsistent comparator groups.
Animal Data
Empagliflozin: Empagliflozin dosed directly to juvenile rats from postnatal day (PND) 21 until PND 90 at doses of 1, 10, 30, and 100 mg/kg/day caused increased kidney weights and renal tubular and pelvic dilatation at 100 mg/kg/day, which approximates 13-times the maximum clinical dose of 25 mg, based on AUC. These findings were not observed after a 13-week drug-free, recovery period. These outcomes occurred with drug exposure during periods of renal development in rats that correspond to the late second and third trimester of human renal development.
In embryo-fetal development studies in rats and rabbits, empagliflozin was administered for intervals coinciding with the first trimester period of organogenesis in humans. Doses up to 300 mg/kg/day, which approximates 48-times (rats) and 128-times (rabbits) the maximum clinical dose of 25 mg (based on AUC), did not result in adverse developmental effects. In rats, at higher doses of empagliflozin causing maternal toxicity, malformations of limb bones increased in fetuses at 700 mg/kg/day or 154-times the 25 mg maximum clinical dose. Empagliflozin crosses the placenta and reaches fetal tissues in rats. In the rabbit, higher doses of empagliflozin resulted in maternal and fetal toxicity at 700 mg/kg/day, or 139-times the 25 mg maximum clinical dose.
In pre- and postnatal development studies in pregnant rats, empagliflozin was administered from gestation day 6 through to lactation day 20 (weaning) at up to 100 mg/kg/day (approximately 16-times the 25 mg maximum clinical dose) without maternal toxicity. Reduced body weight was observed in the offspring at greater than or equal to 30 mg/kg/day (approximately 4-times the 25 mg maximum clinical dose).
Metformin hydrochloride: Metformin hydrochloride did not cause adverse developmental effects when administered to pregnant Sprague Dawley rats and rabbits at up to 600 mg/kg/day during the period of organogenesis. This represents an exposure of approximately 2- and 6-times a clinical dose of 2000 mg, based on body surface area (mg/m2) for rats and rabbits, respectively.
Empagliflozin and Metformin hydrochloride: No adverse developmental effects were observed when empagliflozin and metformin hydrochloride were coadministered to pregnant rats during the period of organogenesis at exposures of approximately 35- and 14-times the clinical AUC exposure of empagliflozin associated with the 10 mg and 25 mg doses, respectively, and 4-times the clinical AUC exposure of metformin associated with the 2000 mg dose.
Risk Summary
There is limited information regarding the presence of SYNJARDY or its components (empagliflozin or metformin) in human milk, the effects on the breastfed infant, or the effects on milk production. Limited published studies report that metformin is present in human milk (see Data). Empagliflozin is present in the milk of lactating rats (see Data). Since human kidney maturation occurs in utero and during the first 2 years of life when lactational exposure may occur, there may be risk to the developing human kidney.
Because of the potential for serious adverse reactions in a breastfed infant, including the potential for empagliflozin to affect postnatal renal development, advise patients that use of SYNJARDY is not recommended while breastfeeding.
Data
Published clinical lactation studies report that metformin is present in human milk which resulted in infant doses approximately 0.11% to 1% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.13 and 1. However, the studies were not designed to definitely establish the risk of use of metformin during lactation because of small sample size and limited adverse event data collected in infants.
Empagliflozin was present at a low level in rat fetal tissues after a single oral dose to the dams at gestation day 18. In rat milk, the mean milk to plasma ratio ranged from 0.634 to 5, and was greater than one from 2 to 24 hours post-dose. The mean maximal milk to plasma ratio of 5 occurred at 8 hours post-dose, suggesting accumulation of empagliflozin in the milk. Juvenile rats directly exposed to empagliflozin showed a risk to the developing kidney (renal pelvic and tubular dilatations) during maturation.
Empagliflozin
In empagliflozin type 2 diabetes studies, 2721 empagliflozin-treated patients were 65 years of age and older and 491 patients were 75 years of age and older. In these studies, volume depletion-related adverse reactions occurred in 2.1%, 2.3%, and 4.4% of patients 75 years of age and older in the placebo, empagliflozin 10 mg, and empagliflozin 25 mg once daily groups, respectively; and urinary tract infections occurred in 10.5%, 15.7%, and 15.1% of patients 75 years of age and older in the placebo, empagliflozin 10 mg, and empagliflozin 25 mg once daily groups, respectively.
Metformin hydrochloride
Clinical studies of metformin did not include sufficient numbers of geriatric patients to determine whether they respond differently from younger adult patients.
Empagliflozin
The glucose lowering benefit of empagliflozin 25 mg decreased in patients with worsening renal function. The risks of renal impairment [see Warnings and Precautions (5.2)], volume depletion adverse reactions and urinary tract infection-related adverse reactions increased with worsening renal function.
Metformin hydrochloride
Metformin is substantially excreted by the kidney, and the risk of metformin accumulation and lactic acidosis increases with the degree of renal impairment [see Warnings and Precautions (5.1)].
Empagliflozin
Empagliflozin is an inhibitor of the sodium-glucose co-transporter 2 (SGLT2).
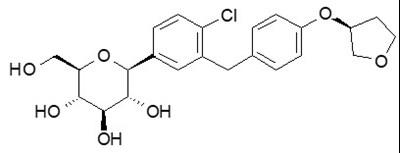
The chemical name of empagliflozin is D-Glucitol,1,5-anhydro-1-C-[4-chloro-3-[[4-[[(3S)-tetrahydro-3-furanyl]oxy]phenyl]methyl]phenyl]-, (1S).
Its molecular formula is C23H27ClO7 and the molecular weight is 450.91. The structural formula is:
Empagliflozin is a white to yellowish, non-hygroscopic powder. It is very slightly soluble in water, sparingly soluble in methanol, slightly soluble in ethanol and acetonitrile; soluble in 50% acetonitrile/water; and practically insoluble in toluene.
Metformin hydrochloride
Metformin hydrochloride (N,N-dimethylimidodicarbonimidic diamide hydrochloride) is a biguanide. Metformin hydrochloride is a white to off-white crystalline compound with a molecular formula of C4H11N5∙HCl and a molecular weight of 165.63. Metformin hydrochloride is freely soluble in water and is practically insoluble in acetone, ether, and chloroform. The pKa of metformin is 12.4. The pH of a 1% aqueous solution of metformin hydrochloride is 6.68. The structural formula is:
SYNJARDY
SYNJARDY tablets for oral administration are available in four dosage strengths containing 5 mg empagliflozin and 500 mg metformin hydrochloride, 5 mg empagliflozin and 1000 mg metformin hydrochloride, 12.5 mg empagliflozin and 500 mg metformin hydrochloride, or 12.5 mg empagliflozin and 1000 mg metformin hydrochloride.
Each film-coated tablet of SYNJARDY contains the following inactive ingredients: copovidone, corn starch, colloidal silicon dioxide, magnesium stearate. Film-coating: hypromellose, titanium dioxide, talc, polyethylene glycol 400, and yellow ferric oxide (5 mg/500 mg, 5 mg/1000 mg) or red ferric oxide and black ferrosoferric oxide (12.5 mg/500 mg, 12.5 mg/1000 mg).
SYNJARDY
SYNJARDY contains: empagliflozin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, and metformin, a biguanide.
Empagliflozin
Sodium-glucose co-transporter 2 (SGLT2) is the predominant transporter responsible for reabsorption of glucose from the glomerular filtrate back into the circulation. Empagliflozin is an inhibitor of SGLT2. By inhibiting SGLT2, empagliflozin reduces renal reabsorption of filtered glucose and lowers the renal threshold for glucose, and thereby increases urinary glucose excretion.
Metformin HCl
Metformin is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes mellitus, lowering both basal and postprandial plasma glucose. It is not chemically or pharmacologically related to any other classes of oral antihyperglycemic agents. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. Unlike SUs, metformin does not produce hypoglycemia in either patients with type 2 diabetes mellitus or normal subjects (except in special circumstances) [see Warnings and Precautions (5.5)] and does not cause hyperinsulinemia. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may actually decrease.
Empagliflozin
Urinary Glucose Excretion
In patients with type 2 diabetes, urinary glucose excretion increased immediately following a dose of empagliflozin and was maintained at the end of a 4-week treatment period averaging at approximately 64 grams per day with 10 mg empagliflozin and 78 grams per day with 25 mg empagliflozin once daily [see Clinical Studies (14)]. Data from single oral doses of empagliflozin in healthy subjects indicate that, on average, the elevation in urinary glucose excretion approaches baseline by about 3 days for the 10 mg and 25 mg doses.
Urinary Volume
In a 5-day study, mean 24-hour urine volume increase from baseline was 341 mL on Day 1 and 135 mL on Day 5 of empagliflozin 25 mg once daily treatment.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, crossover study, 30 healthy subjects were administered a single oral dose of empagliflozin 25 mg, empagliflozin 200 mg (8 times the maximum dose), moxifloxacin, and placebo. No increase in QTc was observed with either 25 mg or 200 mg empagliflozin.
SYNJARDY
Administration of 12.5 mg empagliflozin/1000 mg metformin hydrochloride under fed conditions resulted in a 9% decrease in AUC and a 28% decrease in Cmax for empagliflozin, when compared to fasted conditions. For metformin, AUC decreased by 12% and Cmax decreased by 26% compared to fasting conditions. The observed effect of food on empagliflozin and metformin is not considered to be clinically relevant.
Empagliflozin
Absorption
The pharmacokinetics of empagliflozin has been characterized in healthy volunteers and patients with type 2 diabetes and no clinically relevant differences were noted between the two populations. After oral administration, peak plasma concentrations of empagliflozin were reached at 1.5 hours post-dose. Thereafter, plasma concentrations declined in a biphasic manner with a rapid distribution phase and a relatively slow terminal phase. The steady-state mean plasma AUC and Cmax were 1870 nmol∙h/L and 259 nmol/L, respectively, with 10 mg empagliflozin once daily treatment, and 4740 nmol∙h/L and 687 nmol/L, respectively, with 25 mg empagliflozin once daily treatment. Systemic exposure of empagliflozin increased in a dose-proportional manner in the therapeutic dose range. The single-dose and steady-state pharmacokinetic parameters of empagliflozin were similar, suggesting linear pharmacokinetics with respect to time.
Administration of 25 mg empagliflozin after intake of a high-fat and high-calorie meal resulted in slightly lower exposure; AUC decreased by approximately 16% and Cmax decreased by approximately 37%, compared to fasted condition. The observed effect of food on empagliflozin pharmacokinetics was not considered clinically relevant and empagliflozin may be administered with or without food.
Distribution
The apparent steady-state volume of distribution was estimated to be 73.8 L based on a population pharmacokinetic analysis. Following administration of an oral [14C]-empagliflozin solution to healthy subjects, the red blood cell partitioning was approximately 36.8% and plasma protein binding was 86.2%.
Elimination
The apparent terminal elimination half-life of empagliflozin was estimated to be 12.4 h and apparent oral clearance was 10.6 L/h based on the population pharmacokinetic analysis. Following once daily dosing, up to 22% accumulation, with respect to plasma AUC, was observed at steady-state, which was consistent with empagliflozin half-life.
Metabolism: No major metabolites of empagliflozin were detected in human plasma and the most abundant metabolites were three glucuronide conjugates (2-O-, 3-O-, and 6-O-glucuronide). Systemic exposure of each metabolite was less than 10% of total drug-related material. In vitro studies suggested that the primary route of metabolism of empagliflozin in humans is glucuronidation by the uridine 5'-diphospho-glucuronosyltransferases UGT2B7, UGT1A3, UGT1A8, and UGT1A9.
Excretion: Following administration of an oral [14C]-empagliflozin solution to healthy subjects, approximately 95.6% of the drug-related radioactivity was eliminated in feces (41.2%) or urine (54.4%). The majority of drug-related radioactivity recovered in feces was unchanged parent drug and approximately half of drug-related radioactivity excreted in urine was unchanged parent drug.
Metformin
Absorption
The absolute bioavailability of a metformin hydrochloride 500-mg tablet given under fasting conditions is approximately 50% to 60%. Studies using single oral doses of metformin hydrochloride tablets 500 mg to 1500 mg, and 850 mg to 2550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination.
Food decreases the extent of and slightly delays the absorption of metformin, as shown by approximately a 40% lower Cmax, a 25% lower AUC, and a 35 minute prolongation of time to peak plasma concentration (Tmax) following administration of a single 850 mg tablet of metformin with food, compared to the same tablet strength administered fasting. The clinical relevance of these decreases is unknown.
Distribution
The apparent volume of distribution (V/F) of metformin following single oral doses of immediate-release metformin hydrochloride tablets 850 mg averaged 654±358 L. Metformin is negligibly bound to plasma proteins. Metformin partitions into erythrocytes, most likely as a function of time.
Elimination
Metformin has a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Metabolism: Intravenous single-dose studies in normal subjects demonstrate that metformin does not undergo hepatic metabolism (no metabolites have been identified in humans) nor biliary excretion.
Excretion: Following oral administration, approximately 90% of the absorbed drug is excreted via the renal route within the first 24 hours. Renal clearance is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination.
Specific Populations
Renal Impairment
SYNJARDY: Studies characterizing the pharmacokinetics of empagliflozin and metformin after administration of SYNJARDY in renally impaired patients have not been performed.
Empagliflozin: In patients with mild (eGFR: 60 to less than 90 mL/min/1.73 m2), moderate (eGFR: 30 to less than 60 mL/min/1.73 m2), and severe (eGFR: less than 30 mL/min/1.73 m2) renal impairment and patients with kidney failure/end stage renal disease (ESRD), AUC of empagliflozin increased by approximately 18%, 20%, 66%, and 48%, respectively, compared to subjects with normal renal function. Peak plasma levels of empagliflozin were similar in patients with moderate renal impairment and kidney failure/ESRD, compared to subjects with normal renal function. Peak plasma levels of empagliflozin were roughly 20% higher in patients with mild and severe renal impairment as compared to subjects with normal renal function. Population pharmacokinetic analysis showed that the apparent oral clearance of empagliflozin decreased with a decrease in eGFR leading to an increase in drug exposure. However, the fraction of empagliflozin that was excreted unchanged in urine, and urinary glucose excretion, declined with decrease in eGFR.
Metformin hydrochloride: In patients with decreased renal function, the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased [see Contraindications (4) and Warnings and Precautions (5.1)].
Hepatic Impairment
SYNJARDY: Studies characterizing the pharmacokinetics of empagliflozin and metformin after administration of SYNJARDY in hepatically impaired patients have not been performed [see Warnings and Precautions (5.1)].
Empagliflozin: In patients with mild, moderate, and severe hepatic impairment according to the Child-Pugh classification, AUC of empagliflozin increased by approximately 23%, 47%, and 75%, and Cmax increased by approximately 4%, 23%, and 48%, respectively, compared to subjects with normal hepatic function.
Metformin hydrochloride: No pharmacokinetic studies of metformin have been conducted in patients with hepatic impairment.
Effects of Age, Body Mass Index, Gender, and Race
Empagliflozin: Based on the population PK analysis, age, body mass index (BMI), gender and race (Asians versus primarily Whites) do not have a clinically meaningful effect on pharmacokinetics of empagliflozin [see Use in Specific Populations (8.5)].
Metformin hydrochloride: Metformin pharmacokinetic parameters did not differ significantly between normal subjects and patients with type 2 diabetes mellitus when analyzed according to gender. Similarly, in controlled clinical studies in patients with type 2 diabetes mellitus, the antihyperglycemic effect of metformin was comparable in males and females.
No studies of metformin pharmacokinetic parameters according to race have been performed. In controlled clinical studies of metformin HCl in patients with type 2 diabetes mellitus, the antihyperglycemic effect was comparable in Caucasians (n=249), Blacks (n=51), and Hispanics (n=24).
Geriatric
SYNJARDY: Studies characterizing the pharmacokinetics of empagliflozin and metformin after administration of SYNJARDY in geriatric patients have not been performed [see Warnings and Precautions (5.2) and Use in Specific Populations (8.5)].
Empagliflozin: Age did not have a clinically meaningful impact on the pharmacokinetics of empagliflozin based on a population pharmacokinetic analysis [see Use in Specific Populations (8.5)].
Metformin hydrochloride: Limited data from controlled pharmacokinetic studies of metformin hydrochloride in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and Cmax is increased, compared with healthy young subjects. From these data, it appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function.
Drug Interactions
Pharmacokinetic drug interaction studies with SYNJARDY have not been performed; however, such studies have been conducted with the individual components empagliflozin and metformin HCl.
Empagliflozin
In vitro Assessment of Drug Interactions: Empagliflozin does not inhibit, inactivate, or induce CYP450 isoforms. In vitro data suggest that the primary route of metabolism of empagliflozin in humans is glucuronidation by the uridine 5'-diphospho-glucuronosyltransferases UGT1A3, UGT1A8, UGT1A9, and UGT2B7. Empagliflozin does not inhibit UGT1A1, UGT1A3, UGT1A8, UGT1A9, or UGT2B7. Therefore, no effect of empagliflozin is anticipated on concomitantly administered drugs that are substrates of the major CYP450 isoforms or UGT1A1, UGT1A3, UGT1A8, UGT1A9, or UGT2B7. The effect of UGT induction (e.g., induction by rifampicin or any other UGT enzyme inducer) on empagliflozin exposure has not been evaluated.
Empagliflozin is a substrate for P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP), but it does not inhibit these efflux transporters at therapeutic doses. Based on in vitro studies, empagliflozin is considered unlikely to cause interactions with drugs that are P-gp substrates. Empagliflozin is a substrate of the human uptake transporters OAT3, OATP1B1, and OATP1B3, but not OAT1 and OCT2. Empagliflozin does not inhibit any of these human uptake transporters at clinically relevant plasma concentrations and, therefore, no effect of empagliflozin is anticipated on concomitantly administered drugs that are substrates of these uptake transporters.
In vivo Assessment of Drug Interactions: Empagliflozin pharmacokinetics were similar with and without coadministration of metformin hydrochloride, glimepiride, pioglitazone, sitagliptin, linagliptin, warfarin, verapamil, ramipril, and simvastatin in healthy volunteers and with or without coadministration of hydrochlorothiazide and torsemide in patients with type 2 diabetes (see Figure 1). In subjects with normal renal function, coadministration of empagliflozin with probenecid resulted in a 30% decrease in the fraction of empagliflozin excreted in urine without any effect on 24-hour urinary glucose excretion. The relevance of this observation to patients with renal impairment is unknown.
Figure 1 Effect of Various Medications on the Pharmacokinetics of Empagliflozin as Displayed as 90% Confidence Interval of Geometric Mean AUC and Cmax Ratios [reference lines indicate 100% (80% - 125%)]
| aempagliflozin, 50 mg, once daily; bempagliflozin, 25 mg, single dose; cempagliflozin, 25 mg, once daily; dempagliflozin, 10 mg, single dose |
Empagliflozin had no clinically relevant effect on the pharmacokinetics of metformin, glimepiride, pioglitazone, sitagliptin, linagliptin, warfarin, digoxin, ramipril, simvastatin, hydrochlorothiazide, torsemide, and oral contraceptives when coadministered with empagliflozin (see Figure 2).
Figure 2 Effect of Empagliflozin on the Pharmacokinetics of Various Medications as Displayed as 90% Confidence Interval of Geometric Mean AUC and Cmax Ratios [reference lines indicate 100% (80% - 125%)]
| aempagliflozin, 50 mg, once daily; bempagliflozin, 25 mg, once daily; cempagliflozin, 25 mg, single dose; dadministered as simvastatin; eadministered as warfarin racemic mixture; fadministered as Microgynon®; gadministered as ramipril |
Metformin hydrochloride
| Coadministered Drug | Dose of Coadministered Drug* | Dose of Metformin hydrochloride* | Geometric Mean Ratio (ratio with/without coadministered drug) No effect=1.0 | ||
|---|---|---|---|---|---|
| AUC† | Cmax | ||||
| * All metformin and coadministered drugs were given as single doses | |||||
| † AUC = AUC(INF) | |||||
| ‡ Ratio of arithmetic means | |||||
| **At steady-state with topiramate 100 mg every 12 hours and metformin 500 mg every 12 hours; AUC = AUC(0-12hours) | |||||
| Glyburide | 5 mg | 850 mg | metformin | 0.91‡ | 0.93‡ |
| Furosemide | 40 mg | 850 mg | metformin | 1.09‡ | 1.22‡ |
| Nifedipine | 10 mg | 850 mg | metformin | 1.16 | 1.21 |
| Propranolol | 40 mg | 850 mg | metformin | 0.90 | 0.94 |
| Ibuprofen | 400 mg | 850 mg | metformin | 1.05‡ | 1.07‡ |
| Cationic drugs eliminated by renal tubular secretion may reduce metformin elimination [see Drug Interactions (7)]. | |||||
| Cimetidine | 400 mg | 850 mg | metformin | 1.40 | 1.61 |
| Carbonic anhydrase inhibitors may cause metabolic acidosis [see Drug Interactions (7)]. | |||||
| Topiramate** | 100 mg | 500 mg | metformin | 1.25 | 1.17 |
| Coadministered Drug | Dose of Coadministered Drug* | Dose of Metformin hydrochloride* | Geometric Mean Ratio (ratio with/without metformin) No effect=1.0 | ||
|---|---|---|---|---|---|
| AUC† | Cmax | ||||
| * All metformin and coadministered drugs were given as single doses | |||||
| † AUC = AUC(INF) unless otherwise noted | |||||
| ‡ Ratio of arithmetic means, p-value of difference <0.05 | |||||
| § AUC(0-24 hours) reported | |||||
| ¶ Ratio of arithmetic means | |||||
| Glyburide | 5 mg | 500 mg§ | glyburide | 0.78‡ | 0.63‡ |
| Furosemide | 40 mg | 850 mg | furosemide | 0.87‡ | 0.69‡ |
| Nifedipine | 10 mg | 850 mg | nifedipine | 1.10§ | 1.08 |
| Propranolol | 40 mg | 850 mg | propranolol | 1.01§ | 0.94 |
| Ibuprofen | 400 mg | 850 mg | ibuprofen | 0.97¶ | 1.01¶ |
| Cimetidine | 400 mg | 850 mg | cimetidine | 0.95§ | 1.01 |
SYNJARDY
No carcinogenicity, mutagenicity, or impairment of fertility studies have been conducted with the combination of empagliflozin and metformin HCl. General toxicity studies in rats up to 13 weeks were performed with the combined components. These studies indicated that no additive toxicity is caused by the combination of empagliflozin and metformin.
Empagliflozin
Carcinogenesis was evaluated in 2-year studies conducted in CD-1 mice and Wistar rats. Empagliflozin did not increase the incidence of tumors in female rats dosed at 100, 300, or 700 mg/kg/day (up to 72 times the exposure from the maximum clinical dose of 25 mg). In male rats, hemangiomas of the mesenteric lymph node were increased significantly at 700 mg/kg/day or approximately 42 times the exposure from a 25 mg clinical dose. Empagliflozin did not increase the incidence of tumors in female mice dosed at 100, 300, or 1000 mg/kg/day (up to 62 times the exposure from a 25 mg clinical dose). Renal tubule adenomas and carcinomas were observed in male mice at 1000 mg/kg/day, which is approximately 45 times the exposure of the maximum clinical dose of 25 mg. These tumors may be associated with a metabolic pathway predominantly present in the male mouse kidney.
Empagliflozin was not mutagenic or clastogenic with or without metabolic activation in the in vitro Ames bacterial mutagenicity assay, the in vitro L5178Y tk+/- mouse lymphoma cell assay, and an in vivo micronucleus assay in rats.
Empagliflozin had no effects on mating, fertility or early embryonic development in treated male or female rats up to the high dose of 700 mg/kg/day (approximately 155 times the 25 mg clinical dose in males and females, respectively).
Metformin hydrochloride
Long-term carcinogenicity studies have been performed in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1500 mg/kg/day, respectively. These doses are both approximately 4 times the maximum recommended human daily dose of 2000 mg/kg/day based on body surface area comparisons. No evidence of carcinogenicity with metformin was found in either male or female mice. Similarly, there was no tumorigenic potential observed with metformin in male rats. There was, however, an increased incidence of benign stromal uterine polyps in female rats treated with 900 mg/kg/day.
There was no evidence of a mutagenic potential of metformin in the following in vitro tests: Ames test (Salmonella typhimurium), gene mutation test (mouse lymphoma cells), or chromosomal aberrations test (human lymphocytes). Results in the in vivo mouse micronucleus test were also negative.
Fertility of male or female rats was unaffected by metformin when administered at doses as high as 600 mg/kg/day, which is approximately 2 times the MRHD based on body surface area comparisons.
SYNJARDY Glycemic Control Studies
In patients with type 2 diabetes, treatment with empagliflozin and metformin produced clinically and statistically significant improvements in HbA1c compared to placebo. Reductions in HbA1c were observed across subgroups including age, gender, race, and baseline body mass index (BMI).
Empagliflozin Add-On Combination Therapy with Metformin
A total of 637 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of empagliflozin in combination with metformin.
Patients with type 2 diabetes inadequately controlled on at least 1500 mg of metformin hydrochloride per day entered an open-label 2-week placebo run-in. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10% were randomized to placebo, empagliflozin 10 mg, or empagliflozin 25 mg.
At Week 24, treatment with empagliflozin 10 mg or 25 mg daily provided statistically significant reductions in HbA1c (p-value <0.0001), FPG, and body weight compared with placebo (see Table 7).
| Empagliflozin 10 mg + Metformin N=217 | Empagliflozin 25 mg + Metformin N=213 | Placebo + Metformin N=207 | |
|---|---|---|---|
| aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 9.7%, 14.1%, and 24.6% was imputed for patients randomized to empagliflozin 10 mg, empagliflozin 25 mg, and placebo, respectively. | |||
| bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.) | |||
| cFPG (mg/dL); for empagliflozin 10 mg, n=216, for empagliflozin 25 mg, n=213, and for placebo, n=207 | |||
| HbA1c (%)a | |||
| Baseline (mean) | 7.9 | 7.9 | 7.9 |
| Change from baseline (adjusted mean) | -0.7 | -0.8 | -0.1 |
| Difference from placebo + metformin (adjusted mean) (95% CI) | -0.6b (-0.7, -0.4) | -0.6b (-0.8, -0.5) | -- |
| Patients [n (%)] achieving HbA1c <7% | 75 (38%) | 74 (39%) | 23 (13%) |
| FPG (mg/dL)c | |||
| Baseline (mean) | 155 | 149 | 156 |
| Change from baseline (adjusted mean) | -20 | -22 | 6 |
| Difference from placebo + metformin (adjusted mean) | -26 | -29 | -- |
| Body Weight | |||
| Baseline mean in kg | 82 | 82 | 80 |
| % change from baseline (adjusted mean) | -2.5 | -2.9 | -0.5 |
| Difference from placebo (adjusted mean) (95% CI) | -2.0b (-2.6, -1.4) | -2.5b (-3.1, -1.9) | -- |
At Week 24, the systolic blood pressure was statistically significantly reduced compared to placebo by -4.1 mmHg (placebo-corrected, p-value <0.0001) for empagliflozin 10 mg and -4.8 mmHg (placebo-corrected, p-value <0.0001) for empagliflozin 25 mg.
Empagliflozin Initial Combination Therapy with Metformin
A total of 1364 patients with type 2 diabetes participated in a double-blind, randomized, active-controlled study to evaluate the efficacy and safety of empagliflozin in combination with metformin as initial therapy compared to the corresponding individual components.
Treatment-naïve patients with inadequately controlled type 2 diabetes entered an open-label placebo run-in for 2 weeks. At the end of the run-in period, patients who remained inadequately controlled and had an HbA1c between 7 and 10.5% were randomized to one of 8 active-treatment arms: empagliflozin 10 mg or 25 mg; metformin hydrochloride 1000 mg, or 2000 mg; empagliflozin 10 mg in combination with 1000 mg or 2000 mg metformin hydrochloride; or empagliflozin 25 mg in combination with 1000 mg or 2000 mg metformin hydrochloride.
At Week 24, initial therapy of empagliflozin in combination with metformin provided statistically significant reductions in HbA1c (p-value <0.01) compared to the individual components (see Table 8).
| Empagliflozin 10 mg + Metformin 1000 mga N=161 | Empagliflozin 10 mg + Metformin 2000 mga N=167 | Empagliflozin 25 mg + Metformin 1000 mga N=165 | Empagliflozin 25 mg + Metformin 2000 mga N=169 | Empagliflozin 10 mg N=169 | Empagliflozin 25 mg N=163 | Metformin 1000 mga N=167 | Metformin 2000 mga N=162 | |
|---|---|---|---|---|---|---|---|---|
| aMetformin hydrochloride total daily dose, administered in two equally divided doses per day. | ||||||||
| bp-value ≤0.0062 (modified intent to treat population [observed case] MMRM model included treatment, renal function, region, visit, visit by treatment interaction, and baseline HbA1c). | ||||||||
| cp-value ≤0.0056 (modified intent to treat population [observed case] MMRM model included treatment, renal function, region, visit, visit by treatment interaction, and baseline HbA1c). | ||||||||
| HbA1c (%) | ||||||||
| Baseline (mean) | 8.7 | 8.7 | 8.8 | 8.7 | 8.6 | 8.9 | 8.7 | 8.6 |
| Change from baseline (adjusted mean) | -2.0 | -2.1 | -1.9 | -2.1 | -1.4 | -1.4 | -1.2 | -1.8 |
| Comparison vs empagliflozin (adjusted mean) (95% CI) | -0.6b (-0.9, -0.4) | -0.7b (-1.0, -0.5) | -0.6c (-0.8, -0.3) | -0.7c (-1.0, -0.5) | -- | -- | -- | -- |
| Comparison vs metformin (adjusted mean) (95% CI) | -0.8b (-1.0, -0.6) | -0.3b (-0.6, -0.1) | -0.8c (-1.0, -0.5) | -0.3c (-0.6, -0.1) | -- | -- | -- | -- |
| Patients [n (%)] achieving HbA1c <7% | 96 (63%) | 112 (70%) | 91 (57%) | 111 (68%) | 69 (43%) | 51 (32%) | 63 (38%) | 92 (58%) |
Empagliflozin Add-On Combination Therapy with Metformin and Sulfonylurea
A total of 666 patients with type 2 diabetes participated in a double-blind, placebo-controlled study to evaluate the efficacy and safety of empagliflozin in combination with metformin plus a sulfonylurea.
Patients with inadequately controlled type 2 diabetes on at least 1500 mg per day of metformin hydrochloride and on a sulfonylurea, entered a 2-week open-label placebo run-in. At the end of the run-in, patients who remained inadequately controlled and had an HbA1c between 7% and 10% were randomized to placebo, empagliflozin 10 mg, or empagliflozin 25 mg.
Treatment with empagliflozin 10 mg or 25 mg daily provided statistically significant reductions in HbA1c (p-value <0.0001), FPG, and body weight compared with placebo (see Table 9).
| Empagliflozin 10 mg + Metformin + SU N=225 | Empagliflozin 25 mg + Metformin + SU N=216 | Placebo + Metformin + SU N=225 | |
|---|---|---|---|
| aModified intent to treat population. Last observation on study (LOCF) was used to impute missing data at Week 24. At Week 24, 17.8%, 16.7%, and 25.3% was imputed for patients randomized to empagliflozin 10 mg, empagliflozin 25 mg, and placebo, respectively. | |||
| bANCOVA p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region. Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.) | |||
| cFPG (mg/dL); for empagliflozin 10 mg, n=225, for empagliflozin 25 mg, n=215, for placebo, n=224 | |||
| HbA1c (%)a | |||
| Baseline (mean) | 8.1 | 8.1 | 8.2 |
| Change from baseline (adjusted mean) | -0.8 | -0.8 | -0.2 |
| Difference from placebo (adjusted mean) (95% CI) | -0.6b (-0.8, -0.5) | -0.6b (-0.7, -0.4) | -- |
| Patients [n (%)] achieving HbA1c <7% | 55 (26%) | 65 (32%) | 20 (9%) |
| FPG (mg/dL)c | |||
| Baseline (mean) | 151 | 156 | 152 |
| Change from baseline (adjusted mean) | -23 | -23 | 6 |
| Difference from placebo (adjusted mean) | -29 | -29 | -- |
| Body Weight | |||
| Baseline mean in kg | 77 | 78 | 76 |
| % change from baseline (adjusted mean) | -2.9 | -3.2 | -0.5 |
| Difference from placebo (adjusted mean) (95% CI) | -2.4b (-3.0, -1.8) | -2.7b (-3.3, -2.1) | -- |
Active-Controlled Study vs Glimepiride in Combination with Metformin
The efficacy of empagliflozin was evaluated in a double-blind, glimepiride-controlled, study in 1545 patients with type 2 diabetes with insufficient glycemic control despite metformin therapy.
Patients with inadequate glycemic control and an HbA1c between 7% and 10% after a 2-week run-in period were randomized to glimepiride or empagliflozin 25 mg.
At Week 52, empagliflozin 25 mg and glimepiride lowered HbA1c and FPG (see Table 10, Figure 3). The difference in observed effect size between empagliflozin 25 mg and glimepiride excluded the pre-specified non-inferiority margin of 0.3%. The mean daily dose of glimepiride was 2.7 mg and the maximal approved dose in the United States is 8 mg per day.
| Empagliflozin 25 mg + Metformin N=765 | Glimepiride + Metformin N=780 | |
|---|---|---|
| aModified intent to treat population. Last observation on study (LOCF) was used to impute data missing at Week 52. At Week 52, data was imputed for 15.3% and 21.9% of patients randomized to empagliflozin 25 mg and glimepiride, respectively. | ||
| bNon-inferior, ANCOVA model p-value <0.0001 (HbA1c: ANCOVA model includes baseline HbA1c, treatment, renal function, and region) | ||
| cANCOVA p-value <0.0001 (Body weight and FPG: same model used as for HbA1c but additionally including baseline body weight/baseline FPG, respectively.) | ||
| dFPG (mg/dL); for empagliflozin 25 mg, n=764, for glimepiride, n=779 | ||
| HbA1c (%)a | ||
| Baseline (mean) | 7.9 | 7.9 |
| Change from baseline (adjusted mean) | -0.7 | -0.7 |
| Difference from glimepiride (adjusted mean) (97.5% CI) | -0.07b (-0.15, 0.01) | -- |
| FPG (mg/dL)d | ||
| Baseline (mean) | 150 | 150 |
| Change from baseline (adjusted mean) | -19 | -9 |
| Difference from glimepiride (adjusted mean) | -11 | -- |
| Body Weight | ||
| Baseline mean in kg | 82.5 | 83 |
| % change from baseline (adjusted mean) | -3.9 | 2.0 |
| Difference from glimepiride (adjusted mean) (95% CI) | -5.9c (-6.3, -5.5) | -- |
Figure 3 Adjusted mean HbA1c Change at Each Time Point (Completers) and at Week 52 (mITT Population) - LOCF
At Week 52, the adjusted mean change from baseline in systolic blood pressure was -3.6 mmHg, compared to 2.2 mmHg for glimepiride. The differences between treatment groups for systolic blood pressure was statistically significant (p-value <0.0001).
At Week 104, the adjusted mean change from baseline in HbA1c was -0.75% for empagliflozin 25 mg and -0.66% for glimepiride. The adjusted mean treatment difference was -0.09% with a 97.5% confidence interval of (-0.32%, 0.15%), excluding the pre-specified non-inferiority margin of 0.3%. The mean daily dose of glimepiride was 2.7 mg and the maximal approved dose in the United States is 8 mg per day. The Week 104 analysis included data with and without concomitant glycemic rescue medication, as well as off-treatment data. Missing data for patients not providing any information at the visit were imputed based on the observed off-treatment data. In this multiple imputation analysis, 13.9% of the data were imputed for empagliflozin 25 mg and 12.9% for glimepiride.
At Week 104, empagliflozin 25 mg daily resulted in a statistically significant difference in change from baseline for body weight compared to glimepiride (-3.1 kg for empagliflozin 25 mg vs. +1.3 kg for glimepiride; ANCOVA-LOCF, p-value <0.0001).
Empagliflozin Cardiovascular Outcome Study in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
Empagliflozin is indicated to reduce the risk of cardiovascular death in adults with type 2 diabetes mellitus and established cardiovascular disease. The effect of empagliflozin on cardiovascular risk in adult patients with type 2 diabetes and established, stable, atherosclerotic cardiovascular disease is presented below.
The EMPA-REG OUTCOME study, a multicenter, multi-national, randomized, double-blind parallel group trial compared the risk of experiencing a major adverse cardiovascular event (MACE) between empagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes and atherosclerotic cardiovascular disease. Coadministered antidiabetic medications were to be kept stable for the first 12 weeks of the trial. Thereafter, antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 7020 patients were treated (empagliflozin 10 mg = 2345; empagliflozin 25 mg = 2342; placebo = 2333) and followed for a median of 3.1 years. Approximately 72% of the study population was Caucasian, 22% was Asian, and 5% was Black. The mean age was 63 years and approximately 72% were male.
All patients in the study had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean HbA1c at baseline was 8.1% and 57% of participants had diabetes for more than 10 years. Approximately 31%, 22% and 20% reported a past history of neuropathy, retinopathy and nephropathy to investigators respectively and the mean eGFR was 74 mL/min/1.73 m2. At baseline, patients were treated with one (~30%) or more (~70%) antidiabetic medications including metformin (74%), insulin (48%), and sulfonylurea (43%).
All patients had established atherosclerotic cardiovascular disease at baseline including one (82%) or more (18%) of the following: a documented history of coronary artery disease (76%), stroke (23%) or peripheral artery disease (21%). At baseline, the mean systolic blood pressure was 136 mmHg, the mean diastolic blood pressure was 76 mmHg, the mean LDL was 86 mg/dL, the mean HDL was 44 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 175 mg/g. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 65% with beta-blockers, 43% with diuretics, 77% with statins, and 86% with antiplatelet agents (mostly aspirin).
The primary endpoint in EMPA-REG OUTCOME was the time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event was defined as occurrence of either a cardiovascular death or a non-fatal myocardial infarction (MI) or a non-fatal stroke. The statistical analysis plan had pre-specified that the 10 and 25 mg doses would be combined. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE and superiority on MACE if non-inferiority was demonstrated. Type-1 error was controlled across multiples tests using a hierarchical testing strategy.
Empagliflozin significantly reduced the risk of first occurrence of primary composite endpoint of cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke (HR: 0.86; 95% CI: 0.74, 0.99). The treatment effect was due to a significant reduction in the risk of cardiovascular death in subjects randomized to empagliflozin (HR: 0.62; 95% CI: 0.49, 0.77), with no change in the risk of non-fatal myocardial infarction or non-fatal stroke (see Table 11 and Figures 4 and 5). Results for the 10 mg and 25 mg empagliflozin doses were consistent with results for the combined dose groups.
| Placebo N=2333 | Empagliflozin N=4687 | Hazard ratio vs placebo (95% CI) | |
|---|---|---|---|
| aTreated set (patients who had received at least one dose of study drug) | |||
| bp−value for superiority (2−sided) 0.04 | |||
| cTotal number of events | |||
| Composite of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke (time to first occurrence)b | 282 (12.1%) | 490 (10.5%) | 0.86 (0.74, 0.99) |
| Non-fatal myocardial infarctionc | 121 (5.2%) | 213 (4.5%) | 0.87 (0.70, 1.09) |
| Non-fatal strokec | 60 (2.6%) | 150 (3.2%) | 1.24 (0.92, 1.67) |
| Cardiovascular deathc | 137 (5.9%) | 172 (3.7%) | 0.62 (0.49, 0.77) |
Figure 4 Estimated Cumulative Incidence of First MACE
Figure 5 Estimated Cumulative Incidence of Cardiovascular Death
The efficacy of empagliflozin on cardiovascular death was generally consistent across major demographic and disease subgroups.
Vital status was obtained for 99.2% of subjects in the trial. A total of 463 deaths were recorded during the EMPA-REG OUTCOME trial. Most of these deaths were categorized as cardiovascular deaths. The non-cardiovascular deaths were only a small proportion of deaths, and were balanced between the treatment groups (2.1% in patients treated with empagliflozin, and 2.4% of patients treated with placebo).
Lactic Acidosis
Inform patients of the risks of lactic acidosis due to metformin, its symptoms, and conditions that predispose to its development. Advise patients to discontinue SYNJARDY immediately and to notify their doctor promptly if unexplained hyperventilation, malaise, myalgia, unusual somnolence, or other nonspecific symptoms occur. Counsel patients against excessive alcohol intake and inform patients about the importance of regular testing of renal function while receiving SYNJARDY. Instruct patients to inform their doctor that they are taking SYNJARDY prior to any surgical or radiological procedure, as temporary discontinuation may be required until renal function has been confirmed to be normal [see Warnings and Precautions (5.1)].
Ketoacidosis
Inform patients that ketoacidosis is a serious life-threatening condition and that cases of ketoacidosis have been reported during use of empagliflozin, sometimes associated with illness or surgery among other risk factors. Instruct patients to check ketones (when possible) if symptoms consistent with ketoacidosis occur even if blood glucose is not elevated. If symptoms of ketoacidosis (including nausea, vomiting, abdominal pain, tiredness, and labored breathing) occur, instruct patients to discontinue SYNJARDY and seek medical attention immediately [see Warnings and Precautions (5.2)].
Volume Depletion
Inform patients that symptomatic hypotension may occur with SYNJARDY and advise them to contact their healthcare provider if they experience such symptoms [see Warnings and Precautions (5.3)]. Inform patients that dehydration may increase the risk for hypotension, and to maintain adequate fluid intake.
Serious Urinary Tract Infections
Inform patients of the potential for urinary tract infections, which may be serious. Provide them with information on the symptoms of urinary tract infections. Advise them to seek medical advice if such symptoms occur [see Warnings and Precautions (5.4)].
Hypoglycemia
Inform patients that the risk of hypoglycemia is increased when SYNJARDY is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin, and that a lower dose of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia [see Warnings and Precautions (5.5)].
Necrotizing Fasciitis of the Perineum (Fournier's Gangrene)
Inform patients that necrotizing infections of the perineum (Fournier's gangrene) have occurred with empagliflozin, a component of SYNJARDY. Counsel patients to promptly seek medical attention if they develop pain or tenderness, redness, or swelling of the genitals or the area from the genitals back to the rectum, along with a fever above 100.4°F or malaise [see Warnings and Precautions (5.6)].
Genital Mycotic Infections in Females (e.g., Vulvovaginitis)
Inform female patients that vaginal yeast infections may occur and provide them with information on the signs and symptoms of vaginal yeast infections. Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.7)].
Genital Mycotic Infections in Males (e.g., Balanitis or Balanoposthitis)
Inform male patients that yeast infection of penis (e.g., balanitis or balanoposthitis) may occur, especially in uncircumcised males and patients with chronic and recurrent infections. Provide them with information on the signs and symptoms of balanitis and balanoposthitis (rash or redness of the glans or foreskin of the penis). Advise them of treatment options and when to seek medical advice [see Warnings and Precautions (5.7)].
Hypersensitivity Reactions
Inform patients that serious hypersensitivity reactions, such as urticaria and angioedema, have been reported with empagliflozin, a component of SYNJARDY. Advise patients to report immediately any skin reaction or angioedema, and to discontinue the drug until they have consulted prescribing physician [see Warnings and Precautions (5.8)].
Vitamin B12 Deficiency
Inform patients about importance of regular hematological parameters while receiving SYNJARDY [see Warnings and Precautions (5.9)].
Laboratory Tests
Inform patients that elevated glucose in urinalysis is expected when taking SYNJARDY.
Pregnancy
Advise pregnant patients, and patients of reproductive potential, of the potential risk to a fetus with treatment with SYNJARDY [see Use in Specific Populations (8.1)]. Instruct patients to report pregnancies to their physicians as soon as possible.
Lactation
Advise patients that breastfeeding is not recommended during treatment with SYNJARDY [see Use in Specific Populations (8.2)].
Patients of Reproductive Potential
Inform patients that treatment with metformin may result in ovulation in some premenopausal anovulatory patients, which may lead to unintended pregnancy [see Use in Specific Populations (8.3)].
Missed Dose
Instruct patients to take SYNJARDY only as prescribed. If a dose is missed, it should be taken as soon as the patient remembers. Advise patients not to double their next dose.
Distributed by:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877 USA
Marketed by:
Boehringer Ingelheim Pharmaceuticals, Inc.
Ridgefield, CT 06877 USA
and
Eli Lilly and Company
Indianapolis, IN 46285 USA
SYNJARDY is a registered trademark of and used under license from Boehringer Ingelheim International GmbH.
Boehringer Ingelheim Pharmaceuticals, Inc. either owns or uses the Jardiance® and EMPA-REG OUTCOME® trademarks under license.
The other brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc.
Copyright © 2021 Boehringer Ingelheim International GmbH
ALL RIGHTS RESERVED
COL8974AF142021
SPL8975A
2.1 Prior To Initiation Of Synjardy
- Assess renal function before initiating SYNJARDY and as clinically indicated [see Warnings and Precautions (5.1, 5.3)].
- In patients with volume depletion, correct this condition before initiating SYNJARDY [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
2.2 Recommended Dosage And Administration
- Individualize the starting dose of SYNJARDY based on the patient's current regimen:
- In patients on metformin HCl, switch to SYNJARDY containing empagliflozin 5 mg with a similar total daily dose of metformin HCl;
- In patients on empagliflozin, switch to SYNJARDY containing metformin HCl 500 mg with a similar total daily dose of empagliflozin;
- In patients already treated with empagliflozin and metformin HCl, switch to SYNJARDY containing the same total daily doses of each component.
- Monitor effectiveness and tolerability, and adjust dosing as appropriate, not to exceed the maximum recommended daily dose of empagliflozin 25 mg and metformin HCl 2000 mg.
- Take SYNJARDY twice daily with meals; with gradual dose escalation to reduce the gastrointestinal side effects due to metformin.
2.3 Dosage Recommendations In Patients With Renal Impairment
- Initiation of SYNJARDY is not recommended in patients with an eGFR less than 45 mL/min/1.73 m2, due to the metformin component.
- SYNJARDY is contraindicated in patients with an eGFR less than 30 mL/min/1.73 m2 or in patients on dialysis [see Contraindications (4), Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
2.4 Discontinuation For Iodinated Contrast Imaging Procedures
Discontinue SYNJARDY at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR less than 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart SYNJARDY if renal function is stable [see Warnings and Precautions (5.1)].
3 Dosage Forms And Strengths
SYNJARDY Tablets:
| Empagliflozin Strength | Metformin HCl Strength | Color/Shape | Tablet Markings |
|---|---|---|---|
| 5 mg | 500 mg | orange yellow, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S5" debossed on one side; the other side is debossed with "500" |
| 5 mg | 1000 mg | brownish yellow, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S5" debossed on one side; the other side is debossed with "1000" |
| 12.5 mg | 500 mg | pale brownish purple, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S12" debossed on one side; the other side is debossed with "500" |
| 12.5 mg | 1000 mg | dark brownish purple, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S12" debossed on one side; the other side is debossed with "1000" |
4 Contraindications
SYNJARDY is contraindicated in patients with:
- Severe renal impairment (eGFR less than 30 mL/min/1.73 m2), end stage renal disease, or dialysis [see Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis [see Warnings and Precautions (5.1)].
- Hypersensitivity to empagliflozin, metformin or any of the excipients in SYNJARDY, reactions such as angioedema have occurred [see Warnings and Precautions (5.8)].
5.1 Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension, and resistant bradyarrhythmias have occurred with severe acidosis. Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate:pyruvate ratio; metformin plasma levels generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of SYNJARDY. In SYNJARDY-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin is dialyzable, with a clearance of up to 170 mL/minute under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue SYNJARDY and report these symptoms to their healthcare provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
5.2 Ketoacidosis
Reports of ketoacidosis, a serious life-threatening condition requiring urgent hospitalization have been identified in clinical trials and postmarketing surveillance in patients with type 1 and type 2 diabetes mellitus receiving sodium glucose co-transporter-2 (SGLT2) inhibitors, including empagliflozin. Fatal cases of ketoacidosis have been reported in patients taking empagliflozin. In placebo-controlled trials of patients with type 1 diabetes, the risk of ketoacidosis was increased in patients who received SGLT2 inhibitors compared to patients who received placebo. SYNJARDY is not indicated for the treatment of patients with type 1 diabetes mellitus [see Indications and Usage (1)].
Patients treated with SYNJARDY who present with signs and symptoms consistent with severe metabolic acidosis should be assessed for ketoacidosis regardless of presenting blood glucose levels, as ketoacidosis associated with SYNJARDY may be present even if blood glucose levels are less than 250 mg/dL. If ketoacidosis is suspected, SYNJARDY should be discontinued, patient should be evaluated, and prompt treatment should be instituted. Treatment of ketoacidosis may require insulin, fluid and carbohydrate replacement.
In many of the postmarketing reports, and particularly in patients with type 1 diabetes, the presence of ketoacidosis was not immediately recognized and institution of treatment was delayed because presenting blood glucose levels were below those typically expected for diabetic ketoacidosis (often less than 250 mg/dL). Signs and symptoms at presentation were consistent with dehydration and severe metabolic acidosis and included nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. In some but not all cases, factors predisposing to ketoacidosis such as insulin dose reduction, acute febrile illness, reduced caloric intake, surgery, pancreatic disorders suggesting insulin deficiency (e.g., type 1 diabetes, history of pancreatitis or pancreatic surgery), and alcohol abuse were identified.
Before initiating SYNJARDY, consider factors in the patient history that may predispose to ketoacidosis including pancreatic insulin deficiency from any cause, caloric restriction, and alcohol abuse.
For patients who undergo scheduled surgery, consider temporarily discontinuing SYNJARDY for at least 3 days prior to surgery [see Clinical Pharmacology (12.2, 12.3)].
Consider monitoring for ketoacidosis and temporarily discontinuing SYNJARDY in other clinical situations known to predispose to ketoacidosis (e.g., prolonged fasting due to acute illness or post-surgery). Ensure risk factors for ketoacidosis are resolved prior to restarting SYNJARDY.
Educate patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue SYNJARDY and seek medical attention immediately if signs and symptoms occur.
5.3 Volume Depletion
Empagliflozin can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)]. There have been post-marketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating SYNJARDY in patients with one or more of these characteristics, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating SYNJARDY. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy.
5.4 Urosepsis And Pyelonephritis
There have been postmarketing reports of serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization in patients receiving SGLT2 inhibitors, including empagliflozin. Treatment with SGLT2 inhibitors increases the risk for urinary tract infections. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly, if indicated [see Adverse Reactions (6)].
5.5 Hypoglycemia With Concomitant Use With Insulin And Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. The risk of hypoglycemia is increased when empagliflozin is used in combination with insulin secretagogues (e.g., sulfonylurea) or insulin [see Adverse Reactions (6.1)]. Metformin may increase the risk of hypoglycemia when combined with insulin and/or an insulin secretagogue. Therefore, a lower dose of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with SYNJARDY.
5.6 Necrotizing Fasciitis Of The Perineum (Fournier's Gangrene)
Reports of necrotizing fasciitis of the perineum (Fournier's gangrene), a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in postmarketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin. Cases have been reported in both females and males. Serious outcomes have included hospitalization, multiple surgeries, and death.
Patients treated with SYNJARDY presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, should be assessed for necrotizing fasciitis. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue SYNJARDY, closely monitor blood glucose levels, and provide appropriate alternative therapy for glycemic control.
5.7 Genital Mycotic Infections
Empagliflozin increases the risk for genital mycotic infections [see Adverse Reactions (6.1)]. Patients with a history of chronic or recurrent genital mycotic infections were more likely to develop genital mycotic infections. Monitor and treat as appropriate.
5.8 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions, (e.g., angioedema) in patients treated with empagliflozin. If a hypersensitivity reaction occurs, discontinue SYNJARDY; treat promptly per standard of care, and monitor until signs and symptoms resolve. SYNJARDY is contraindicated in patients with hypersensitivity to empagliflozin or any of the excipients in SYNJARDY [see Contraindications (4)].
5.9 Vitamin B12 Deficiency
In metformin clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of metformin-treated patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 at 2 to 3 year intervals in patients on SYNJARDY and manage any abnormalities [see Adverse Reactions (6.1)].
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
- Lactic Acidosis [see Boxed Warning and Warnings and Precautions (5.1)]
- Ketoacidosis [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Urosepsis and Pyelonephritis [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Necrotizing Fasciitis of the Perineum (Fournier's Gangrene) [see Warnings and Precautions (5.6)]
- Genital Mycotic Infections [see Warnings and Precautions (5.7)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.8)]
- Vitamin B12 Deficiency [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of concomitantly administered empagliflozin (daily dose 10 mg and 25 mg) and metformin hydrochloride (mean daily dose of approximately 1800 mg) has been evaluated in 3456 patients with type 2 diabetes mellitus treated for 16 to 24 weeks, of which 926 patients received placebo, 1271 patients received a daily dose of empagliflozin 10 mg, and 1259 patients received a daily dose of empagliflozin 25 mg. Discontinuation of therapy due to adverse events across treatment groups was 3.0%, 2.8%, and 2.9% for placebo, empagliflozin 10 mg, and empagliflozin 25 mg, respectively.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
7 Drug Interactions
| Carbonic Anhydrase Inhibitors | |
| Clinical Impact | Topiramate or other carbonic anhydrase inhibitors (e.g., zonisamide, acetazolamide or dichlorphenamide) frequently causes a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. |
| Intervention | Concomitant use of these drugs with SYNJARDY may increase the risk of lactic acidosis. Consider more frequent monitoring of these patients. |
| Drugs that Reduce Metformin Clearance | |
| Clinical Impact | Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir, and cimetidine) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3)]. |
| Intervention | Consider the benefits and risks of concomitant use. |
| Alcohol | |
| Clinical Impact | Alcohol is known to potentiate the effect of metformin on lactate metabolism. |
| Intervention | Warn patients against excessive alcohol intake while receiving SYNJARDY. |
| Diuretics | |
| Clinical Impact | Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. |
| Intervention | Before initiating SYNJARDY, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating SYNJARDY. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. |
| Insulin or Insulin Secretagogues | |
| Clinical Impact | The risk of hypoglycemia is increased when empagliflozin is used in combination with insulin secretagogues (e.g., sulfonylurea) or insulin. Metformin may increase the risk of hypoglycemia when combined with insulin and/or an insulin secretagogue. |
| Intervention | Coadministration of SYNJARDY with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower doses of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
| Drugs Affecting Glycemic Control | |
| Clinical Impact | Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. These drugs include the thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blocking drugs, and isoniazid. |
| Intervention | When such drugs are administered to a patient receiving SYNJARDY, the patient should be closely observed to maintain adequate glycemic control. When such drugs are withdrawn from a patient receiving SYNJARDY, the patient should be observed closely for hypoglycemia. |
| Positive Urine Glucose Test | |
| Clinical Impact | SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
| Intervention | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention | Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control. |
8.3 Females And Males Of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with metformin may result in ovulation in some anovulatory women.
8.4 Pediatric Use
Safety and effectiveness of SYNJARDY have not been established in pediatric patients.
8.5 Geriatric Use
Because renal function abnormalities can occur after initiating empagliflozin, metformin is substantially excreted by the kidney, and aging can be associated with reduced renal function, renal function should be assessed more frequently in elderly patients [see Dosage and Administration (2.1) and Warnings and Precautions (5.1, 5.3)].
8.6 Renal Impairment
SYNJARDY should not be initiated in patients with an eGFR less than 45 mL/min/1.73 m2 due to the metformin component and is contraindicated in patients with severe renal impairment (eGFR less than 30 mL/min/1.73 m2), end stage renal disease, or dialysis.
8.7 Hepatic Impairment
Use of metformin hydrochloride in patients with hepatic impairment has been associated with some cases of lactic acidosis. SYNJARDY is not recommended in patients with hepatic impairment [see Warnings and Precautions (5.1)].
10 Overdosage
In the event of an overdose with SYNJARDY, contact the Poison Control Center.
Overdose of metformin HCl has occurred, including ingestion of amounts greater than 50 grams. Lactic acidosis has been reported in approximately 32% of metformin overdose cases [see Warnings and Precautions (5.1)]. Metformin is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions. Therefore, hemodialysis may be useful for removal of accumulated drug from patients in whom metformin overdosage is suspected.
Removal of empagliflozin by hemodialysis has not been studied.
11 Description
SYNJARDY tablets for oral use contain: empagliflozin and metformin hydrochloride.
16 How Supplied/Storage And Handling
SYNJARDY tablets are available as follows:
| Tablet Strength | Color/Shape | Tablet Markings | Package Size | NDC Number |
|---|---|---|---|---|
| 5 mg Empagliflozin 500 mg Metformin HCl | orange yellow, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S5" debossed on one side; the other side is debossed with "500" | Bottles of 60 Bottles of 180 | 0597-0159-60 0597-0159-18 |
| 5 mg Empagliflozin 1000 mg Metformin HCl | brownish yellow, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S5" debossed on one side; the other side is debossed with "1000" | Bottles of 60 Bottles of 180 | 0597-0175-60 0597-0175-18 |
| 12.5 mg Empagliflozin 500 mg Metformin HCl | pale brownish purple, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S12" debossed on one side; the other side is debossed with "500" | Bottles of 60 Bottles of 180 | 0597-0180-60 0597-0180-18 |
| 12.5 mg Empagliflozin 1000 mg Metformin HCl | dark brownish purple, oval, biconvex, film-coated tablet | Boehringer Ingelheim company symbol and "S12" debossed on one side; the other side is debossed with "1000" | Bottles of 60 Bottles of 180 | 0597-0168-60 0597-0168-18 |
Storage And Handling
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Spl Medguide
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: June 2021 | |||
| MEDICATION GUIDE SYNJARDY® (sin-JAR-dee) (empagliflozin and metformin hydrochloride tablets) for oral use | ||||
| What is the most important information I should know about SYNJARDY? SYNJARDY can cause serious side effects, including:
| ||||
|
| |||
You have a higher chance of getting lactic acidosis with SYNJARDY if you:
| ||||
| ||||
|
| |||
| If you get any of these symptoms during treatment with SYNJARDY, if possible, check for ketones in your urine, even if your blood sugar is less than 250 mg/dL. | ||||
| ||||
|
| |||
| Talk to your doctor about what you can do to prevent dehydration including how much fluid you should drink on a daily basis. Talk to your doctor right away if you reduce the amount of food or liquid you drink, for example if you are sick or cannot eat, or start to lose liquids from your body, for example from vomiting, diarrhea or being in the sun too long. | ||||
| What is SYNJARDY? SYNJARDY is a prescription medicine that contains 2 diabetes medicines, empagliflozin (JARDIANCE) and metformin hydrochloride. SYNJARDY can be used: | ||||
| ||||
| ||||
| Who should not take SYNJARDY? Do not take SYNJARDY if you:
| ||||
| What should I tell my doctor before taking SYNJARDY? Before taking SYNJARDY, tell your doctor about all of your medical conditions, including if you:
SYNJARDY may affect the way other medicines work, and other medicines may affect how SYNJARDY works. Especially tell your doctor if you take: | ||||
How should I take SYNJARDY?
| ||||
| What should I avoid while taking SYNJARDY? Avoid drinking alcohol very often, or drinking a lot of alcohol in a short period of time ("binge" drinking). It can increase your chances of getting serious side effects. | ||||
| What are the possible side effects of SYNJARDY? SYNJARDY may cause serious side effects, including:
| ||||
|
|
|
| |
| ||||
|
|
| ||
| ||||
|
| |||
| These are not all the possible side effects of SYNJARDY. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
How should I store SYNJARDY?
| ||||
| General information about the safe and effective use of SYNJARDY. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use SYNJARDY for a condition for which it was not prescribed. Do not give SYNJARDY to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about SYNJARDY that is written for health professionals. | ||||
| What are the ingredients in SYNJARDY? Active Ingredients: empagliflozin and metformin hydrochloride Inactive Ingredients: copovidone, corn starch, colloidal silicon dioxide, magnesium stearate. In addition, the film coating contains the following inactive ingredients: hypromellose, titanium dioxide, talc, and polyethylene glycol 400. 5 mg/500 mg and 5 mg/1000 mg tablets also contain yellow ferric oxide. 12.5 mg/500 mg and 12.5 mg/1000 mg tablets also contain red ferric oxide and black ferrosoferric oxide | ||||
Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc.; Ridgefield, CT 06877 USA | ||||
Principal Display Panel - 5 Mg/500 Mg Tablet Bottle Label
NDC 0597-0159-60
DISPENSE WITH ACCOMPANYING
MEDICATION GUIDE
Synjardy®
(empagliflozin and
metformin
hydrochloride tablets)
5 mg/500 mg
Rx only
60 tablets
Boehringer
Ingelheim
Lilly
Principal Display Panel - 5 Mg/1000 Mg Tablet Bottle Label
NDC 0597-0175-60
DISPENSE WITH ACCOMPANYING
MEDICATION GUIDE
Synjardy®
(empagliflozin and
metformin
hydrochloride tablets)
5 mg/1000 mg
Rx only
60 tablets
Boehringer
Ingelheim
Lilly
Principal Display Panel - 12.5 Mg/500 Mg Tablet Bottle Label
NDC 0597-0180-60
DISPENSE WITH ACCOMPANYING
MEDICATION GUIDE
Synjardy®
(empagliflozin and
metformin
hydrochloride tablets)
12.5 mg/500 mg
Rx only
60 tablets
Boehringer
Ingelheim
Lilly
Principal Display Panel - 12.5 Mg/1000 Mg Tablet Bottle Label
NDC 0597-0168-60
DISPENSE WITH ACCOMPANYING
MEDICATION GUIDE
Synjardy®
(empagliflozin and
metformin
hydrochloride tablets)
12.5 mg/1000 mg
Rx only
60 tablets
Boehringer
Ingelheim
Lilly
* Please review the disclaimer below.