Product Images Striverdi Respimat
View Photos of Packaging, Labels & Appearance
- striverdi respimat 01
- striverdi respimat 02
- striverdi respimat 03
- striverdi respimat 04
- striverdi respimat 05
- striverdi respimat 06
- striverdi respimat 07
- striverdi respimat 08
- striverdi respimat 09
- striverdi respimat 10
- striverdi respimat 11
- striverdi respimat 12
- striverdi respimat 13
- striverdi respimat 14
- striverdi respimat 15
- striverdi respimat 16
- striverdi respimat 17
- striverdi respimat 18
- striverdi respimat 19
Product Label Images
The following 19 images provide visual information about the product associated with Striverdi Respimat NDC 0597-0192 by Boehringer Ingelheim Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
striverdi respimat 02

This text contains data related to a clinical trial involving four different doses of Olodaterol (2ug, 5ug, 10ug, and 20ug) administered daily to a total of 326 participants. The data recorded includes the average FEV (forced expiratory volume) before and after the treatment, with the number of participants (n) cited for each dose.*
striverdi respimat 03

This appears to be a graph showing the comparison of FEV (Forced Expiratory Volume) between two groups, one receiving a medication called "olodaterol" and the other receiving a placebo, over a period of 8 hours. The values on the Y-axis range from 110 to 160, and the time period on the X-axis runs from 0 to 8 hours after medication dosing. However, the output may not be accurate enough to make a definitive interpretation.*
striverdi respimat 07

The text is describing two separate items. The first item is a "safety catch", which is not further described or defined. The second item is a "clear base", again with no further description or definition. No context is given for these items, so it is not possible to determine their purpose or significance.*
striverdi respimat 19
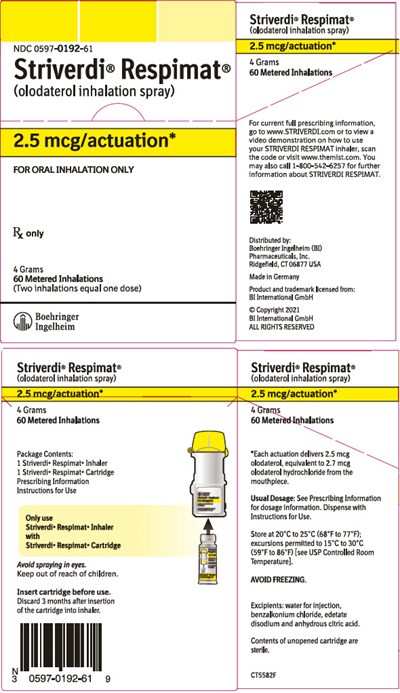
Striverdic Respimate is an olodaterol inhalation spray with an NDC of 0597-0192-61. It is designed for oral inhalation only and comes in a package that contains 4 grams of the product with 60 metered inhalations. The product should only be used as per the instructions provided by the manufacturers. A video demonstration on the correct usage of the STRIVERDI RESPIMAT inhaler is available on the manufacturer's website or by scanning the provided code. The product is made in Germany and is distributed by Boehringer Ingelheim Pharmaceuticals, Inc. The cartridge needs to be inserted before use, and the product needs to be discarded three months after insertion. Apart from these instructions, no other useful information is available due to the presence of unclear text resulting from .*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.














