Product Images Pradaxa
View Photos of Packaging, Labels & Appearance
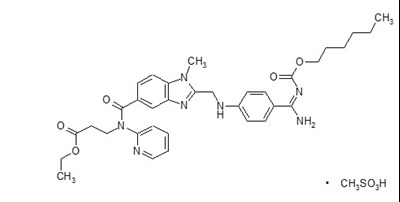
- Chemical Structure - pradaxa 01
- Figure 1 - pradaxa 02
- Figure 2 - pradaxa 03
- Figure - pradaxa 04
- Figure - pradaxa 05
- Figure - pradaxa 06
- Figure - pradaxa 07
- Figure - pradaxa 08
- Figure - pradaxa 09
- Figure - pradaxa 10
- Figure - pradaxa 11
- Figure - pradaxa 12
- Figure - pradaxa 13
- Figure - pradaxa 14
- Figure - pradaxa 15
- Figure - pradaxa 16
- Figure - pradaxa 17
- PRINCIPAL DISPLAY PANEL - 20 mg Packet Bag Carton - pradaxa 18
- PRINCIPAL DISPLAY PANEL - 30 mg Packet Bag Carton - pradaxa 19
- PRINCIPAL DISPLAY PANEL - 40 mg Packet Bag Carton - pradaxa 20
- PRINCIPAL DISPLAY PANEL - 50 mg Packet Bag Carton - pradaxa 21
- PRINCIPAL DISPLAY PANEL - 110 mg Packet Bag Carton - pradaxa 22
- PRINCIPAL DISPLAY PANEL - 150 mg Packet Bag Carton - pradaxa 23
Product Label Images
The following 23 images provide visual information about the product associated with Pradaxa NDC 0597-0445 by Boehringer Ingelheim Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - pradaxa 03
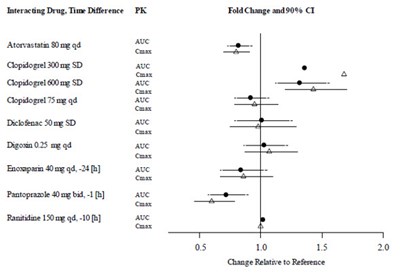
This appears to be a medication list with dosages for different drugs such as Atorvastatin, Clopidogrel and Ranitidine. The list also includes the frequency of intake and abbreviations such as "mg" and "qd" which may refer to specific instructions or dosages. It is unclear what the last section with "Fold Change and 90% CT" is referring to.*
PRINCIPAL DISPLAY PANEL - 20 mg Packet Bag Carton - pradaxa 18

The text provides information about Pradaxa oral pellets, a medication with the active ingredient dabigatran etexilate in 20 mg dose packets. It is advised not to open the packets until ready to use and unused pellets should be discarded 6 months after opening the aluminum bag. The medication should be dispensed in a specific container and cannot be substituted with other dabigatran etexilate forms. The information includes the manufacturer's name and product details.*
PRINCIPAL DISPLAY PANEL - 30 mg Packet Bag Carton - pradaxa 19

This is a product description for Pradaxa oral pellets, a medication containing dabigatran etexilate, dispensed in a unit of use container. The medication guide enclosed provides information on how to use Pradaxa. Each packet contains 30mg of the medication and is not interchangeable with other dosages of dabigatran etexilate. The aluminum bag contains 60 packets and should not be opened until the user is ready to use them. Unused pellets should be discarded after 6 months. The manufacturer is Boehringer Ingelheim.*
PRINCIPAL DISPLAY PANEL - 40 mg Packet Bag Carton - pradaxa 20

This is a description of Pradaxa, a medication used to prevent blood clots in adults who have certain conditions, such as atrial fibrillation. The medication is in the form of oral pellets, containing 40mg per packet. The packaging has a warning not to open the packets until use, and to discard any unused medication after 6 months of opening the aluminum bag. The text also includes the NDC number, caution against substituting with other forms of dabigatran etexilate, and the name of the manufacturer, Boehringer Ingelheim.*
PRINCIPAL DISPLAY PANEL - 50 mg Packet Bag Carton - pradaxa 21

This is a description of Pradaxa, an oral pellet medication containing dabigatran etexilate, which is dispensed in a unit of use container with each packet containing 50 mg. It is essential not to open the packets until usage and discard any unused pellets after six months of opening the aluminum bag. The medication is not interchangeable on a milligram-to-milligram basis with other forms of dabigatran etexilate dosage. The package contains one aluminum bag with 60 packets. There is no other available information in the text.*
PRINCIPAL DISPLAY PANEL - 110 mg Packet Bag Carton - pradaxa 22

NDC 0597-0445-87 is a unit of use container for Pradaxa oral pellets, which should not be substituted on a mg-to-mg basis with other dabigatran etexilate dosage forms. It is important to discard unused PRADAXA oral pellets 6 months after opening the aluminum bag. The product is manufactured by Boehringer.*
PRINCIPAL DISPLAY PANEL - 150 mg Packet Bag Carton - pradaxa 23

This is a description of Pradaxa, an oral medication used to prevent blood clots. It comes in packets containing 150mg of dabigatran etexilate, and should not be substituted with other dosage forms. The packets should not be opened until use, and any unused medication should be discarded six months after opening the aluminum bag. The medication is dispensed in a unit of use container with NDC 0597-0450-16.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.