Product Images Diltiazem Hydrochloride
View Photos of Packaging, Labels & Appearance
- Image - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 1
- Diltiazem ER 180mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 10
- Diltiazem ER 240mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 11
- Image - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 2
- 120 - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 3
- 180 - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 4
- 240 - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 5
- 300 - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 6
- 360 - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 7
- Image - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 8
- Diltiazem ER 120mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 9
Product Label Images
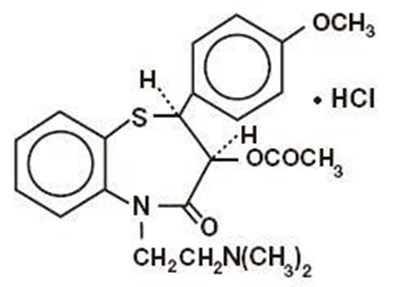
The following 11 images provide visual information about the product associated with Diltiazem Hydrochloride NDC 0615-8380 by Ncs Healthcare Of Ky, Llc Dba Vangard Labs, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Diltiazem ER 180mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 10

This is a product description for Diltiazem HCI ER Caps USP 180mg with a LOT number of 8380. It is manufactured by Novast for Ingenus and distributed by Vangard in Scotland, Kentucky. It is a once-a-day dosage and should be stored at 20-25°C. The packaging is trademarked by Omnicare, Inc. It is for institutional use only and the package insert or label should be consulted for dosage information.*
Diltiazem ER 240mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 11

This is a prescription drug package for Diltiazem HCI ER Caps USP containing 240mg. It is manufactured by Novast for Ingenus and packaged by Vangard in Glasgow, KY. The Novast NDC is 50742.250-90 and Ingenus NDC is 0615-8381-39. The LOT number is 8381, and it must be stored at controlled room temperature. The package insert or label should offer dosage information. This package is to be used for institutional purposes only and dispensed in a light-resistant container.*
Image - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 2
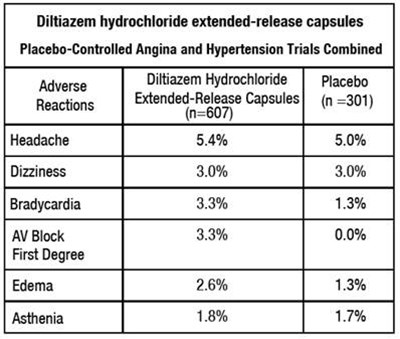
This text appears to be a report on a clinical trial comparing the effects of Diltiazem hydrochloride extended-release capsules versus a placebo on patients with angina and hypertension. The table displays the percentage of patients who experienced adverse reactions in each group, including headache, dizziness, bradycardia, AV block, edema, and asthenia.*
Diltiazem ER 120mg Caps bingo label - diltiazem hcl er caps 120mg 8379 180mg 8380 240mg 9

This is a prescription medication called Diltiazem HCI. It comes in extended-release capsules and each capsule contains either 120mg or 150mg of the active ingredient. The medication is intended for once-a-day dosing. The medication should be stored at room temperature (20°- 25°C or 68°- 77' F) and protected from moisture. It should be dispensed in a light-resistant container. Dosage instructions are available on the package insert or label. The text contains information about different lots and expiration dates of the medication, as well as manufacturing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.