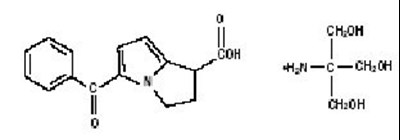
Product Images Ketorolac Tromethamine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Ketorolac Tromethamine NDC 0641-6041 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
15 mg/mL - ketorolac tromethamine injection usp 2

This is a prescription drug labeled as Ketorolac Tromethamine Injection, a pain reliever that can be administered through intravenous or intramuscular methods. Each mL of the injection contains 15 mg of the medicine. The drug is contained in a single-use vial of 1 mL capacity that was manufactured by Hima in Berkeley Heights 07922. The label includes a lot number and an expiration date for quality control purposes.*
15 mg/mL carton - ketorolac tromethamine injection usp 3

This is a label for a medication called Tromethamine Injection, USP. It comes in 25 single-use vials, each containing 15 mg/mL of the medication. It can be administered intravenously or intramuscularly. The label lists the various ingredients and dosage instructions. Storage instructions are also provided.*
Ketorolac Tromethamine Injection, USP 30 mg/mL 1 mL Single Dose Vial - ketorolac tromethamine injection usp 4
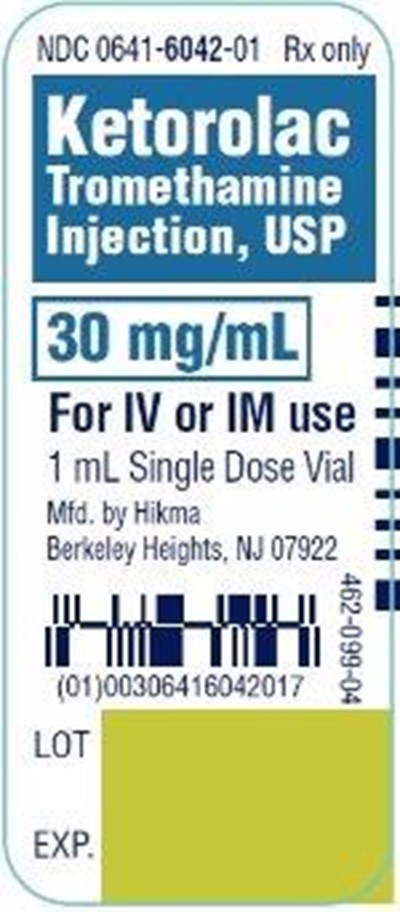
This is a description of a drug called Ketorolac Tromethamine that is available in a 30 mg/mL injection, USP. It can be used for both intravenous (IV) and intramuscular (IM) administration, and comes in a single-dose vial of 1 mL. The drug is manufactured by Hikma and its batch number or lot number is not available in the provided text. The expiration date of the drug is also not available.*
Ketorolac Tromethamine Injection, USP 30 mg/mL 25 x 1 mL Single Dose Vials - ketorolac tromethamine injection usp 5

Tromathamine Injection, USP is a sterile solution for intravenous or intramuscular use. The vial contains 30 mg/mL solution and a single dose is 251 mL. The Ustal Dosage is dependent on the specific medical condition. The solution should be stored between 20-25°C and protected from light.*
Ketorolac Tromethamine Injection, USP 60 mg/2 mL (30 mg/mL) 2 mL Single Dose Vial - ketorolac tromethamine injection usp 6

This is a description of a drug called Tromethamine Injection, USP. It comes in a single dose vial containing 60 mg of medication in 2 ml of liquid. The concentration is 30 mg/mL. The medication should only be administered through an intramuscular injection. The vial is produced by a company called Wi by Ikma located in Beroley Heights, W 07522. There is an NDC number of 0641-6043.01 and an expiration date present on the packaging.*
Ketorolac Tromethamine Injection, USP 60 mg/2 mL (30 mg/mL) 25 x 2 mL Single Dose Vials - ketorolac tromethamine injection usp 7
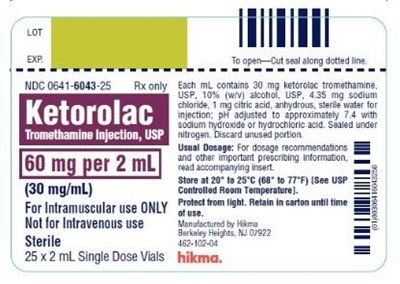
This is a description of a medication called Tromethamine injection, USP, which is sold in a 252 mL single-dose vial. Each vial contains 60 mg of Tromethamine in 2 mL with a recommended dosage for intramuscular use. The medication is sterile and contains 30 mg of Htorolac Tromethamine. The description includes instructions for use, storage, and disposal. The manufacturer is listed as Hikma but the location information is incomplete.*
ketorolac tromethamine injection usp 8

This is a product identification code, which includes a GTIN (Global Trade Item Number), SN (Serial Number), expiration date (EXP) and LOT number. It is not possible to determine what product is associated with this code without additional information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.