Product Images Hydromorphone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 17 images provide visual information about the product associated with Hydromorphone Hydrochloride NDC 0641-6206 by Hikma Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Hydromorphone PFS 0.5 mg Carton - hydromorphone hcl injection usp syringe 11

This is the medicine label for HYDROmorphone HCl Injection, intended for subcutaneous, intramuscular, or slow intravenous use. It contains 0.5 mg of hydromorphone bycroshrids, USP per 0.5 ml. The syringes are for single-dose use and should be discarded after use. The medication should not be used for intrathecal or epidural purposes. The manufacturer is Him Pharmaceuticals USA located in Berkeley Heights, NJ. Storage instructions include protecting from light and keeping at controlled room temperature.*
Hydromorphone PFS 1 mg Carton - hydromorphone hcl injection usp syringe 14

This text appears to be a medication label for Hydromorphone HCI Injection, USP, containing 1 mg per mL for subcutaneous, intramuscular, or slow intravenous use. It warns not to dilute for IV push and not to place the syringe on a sterile field. The medication is also not recommended for spinal or epidural use. The manufacturer is listed as Hikma Pharmaseuticals USA Inc. It provides details on the ingredients and instructions for use.*
Hydromorphone PFS 2 mg Carton - hydromorphone hcl injection usp syringe 17
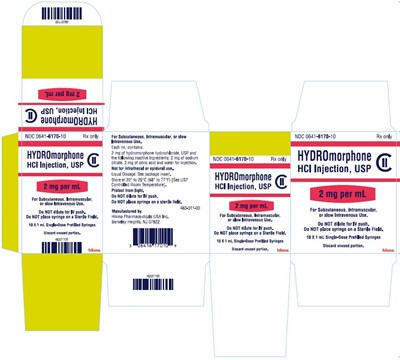
This text appears to be a description of a medication called HYDROmorphone, indicated for subcutaneous, intramuscular, or slow intravenous use. It includes dosage information and instructions for use. The medication is manufactured by a company called Him Pharmaceuticals USA Inc. The text also provides details on the packaging and storage instructions for the medication.*
Hydromorphone PFS 0.2 mg Carton - hydromorphone hcl injection usp syringe 8

This is a product label for HYDROmorphone HCI Injection, USP in 2mg single-dose prefilled syringes for subcutaneous, intramuscular, or slow intravenous use. The usual dosage instructions and storage conditions are provided on the label. Manufactured by Hikma Pharmaceuticals USA in Berkeley Heights, NJ. The label warns not to dilute for IV push and not to place the syringe on a sterile field. Unused portions should be discarded properly.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.












