Product Images Drospirenone/ethinyl Estradiol/levomefolate Calcium And Levomefolate Calcium
View Photos of Packaging, Labels & Appearance
- 50419 0407 01 SPLIMAGE30 42192129
- 50419 0407 01 SPLIMAGE30 44192279
- 52a786c4 4ace 46f0 bda2 2de721b1bb47 00
- Fig 1 - image 01
- Fig 2 - image 02
- Structures - image 03
- Fig 3 - image 04
- Fig 4 - image 05
- Fig 5 - image 06
- Fig 6 - image 07
- birth control chart - image 08
- blister - image 09
- VTE chart - image 10
- carton 3s - image 11
Product Label Images
The following 14 images provide visual information about the product associated with Drospirenone/ethinyl Estradiol/levomefolate Calcium And Levomefolate Calcium NDC 0781-4075 by Sandoz Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
52a786c4 4ace 46f0 bda2 2de721b1bb47 00
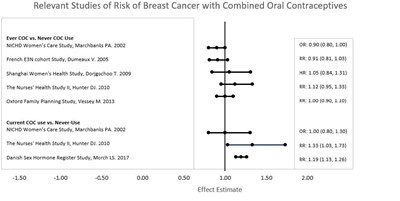
This text presents a list of studies focused on the risk of breast cancer associated with combined oral contraceptives (COC) use. The studies analyzed the risk between ever COC use and never COC use, as well as current COC use and never-use. The text includes some statistical information such as effect estimates, OR, RR, and HR.*
Fig 2 - image 02
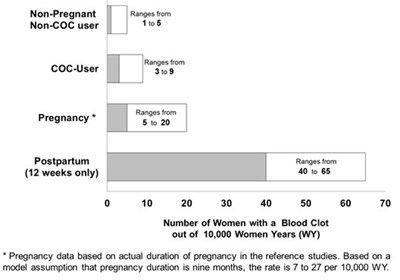
This is a chart displaying the ranges of blood clot occurrences among non-pregnant women, non-COC users, COC users, and postpartum women. The numbers are given out of 10,000 Women Years (WY). There is also a note stating that the pregnancy data is based on actual duration of pregnancy and that the rate of blood clot occurrence is between 7 and 27 per 10,000 WY.*
birth control chart - image 08

This text describes different methods of birth control and their effectiveness rates. The text displays effectiveness rates in terms of pregnancy per 100 women. The text lists methods such as implants, injections, intrauterine devices, sterilization, birth control pills, skin patch, vaginal ring with hormones, condoms, diaphragm, no sex during the most fertile days of the monthly cycle, spermicide, and withdrawal. The most effective methods listed are sterilization and intrauterine devices, while the least effective method listed is no birth control.*
VTE chart - image 10
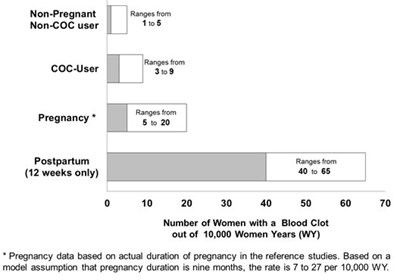
This text provides ranges for non-pregnant women who are either non-COC users or cocusers, as well as ranges for pregnant and postpartum women in terms of the number of women with a blood clot out of 10,000 women years. The pregnancy data is based on actual duration of pregnancy in reference studies, and the rate is assumed to be 7 to 27 per 10,000 WY if pregnancy duration is nine months.*
carton 3s - image 11

Drospirenone/Ethinyl Estradiol/Levomefolate Calcium Tablets and Levomefolate Calcium Tablets with NDC 0781-4075.15 is a product intended for birth control purposes. It comes in 3mg/0.02mg/0.451 mg and 0.451 mg strength and is manufactured by Sandoz. The product is sold in 3 units of 28 oral tablets. The text mentions some symbols, but no further information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.








