Product Images Naloxone Hydrochloride
View Photos of Packaging, Labels & Appearance
- Nalaxone HCl Nasal Spray 4 mg blister label - Blister Label
- Naloxone HCl Nasal Spray 4 mg carton label - Carton Label.JPG
- Naloxone HCl Nasal Spray Quick Start Guide - IFU
- Chemical Structure - narcan 01
- Figure 1(a) - narcan 02
- Figure 1(b) - narcan 03
- Figure - narcan 04
- Figure - narcan 05
- Figure - narcan 06
- Figure - narcan 07
- Figure - narcan 08
Product Label Images
The following 11 images provide visual information about the product associated with Naloxone Hydrochloride NDC 0781-7176 by Sandoz Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Nalaxone HCl Nasal Spray 4 mg blister label - Blister Label

This is a description of Naloxone HCI Nasal Spray, a medication designed to be used in the nose only. It is available by prescription only, and each spray device contains 1 dose of the medication, which contains 4 mg of naloxone HCI, equivalent to 3.6 mg of naloxone, in 0.1 L of nasal spray. The medication is stored below 77°F (25°C), and excursions up to 104°F (40°C) are permitted, but it should not be frozen or exposed to heat above 104°F (40°C). This medication is used for known or suspected opioid overdose in adults and children. The description recommends users to see the enclosed quick start guide and not to test the device before use. The product is distributed by Sandoz, located in Princeton, NJ.*
Figure 1(b) - narcan 03
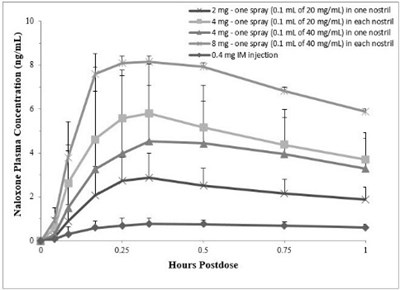
This is a table showing the Naloxone Plasma Concentration (ng/mL) at different time points after administering different doses and routes of Naloxone. The available routes of administration are nasal and intramuscular, and the doses range from 0.2mg to 0.4mg. The time points evaluated are at 0.5 hours and 1 hour post-dose.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.