Product Images Fluvastatin Sodium ER
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Fluvastatin Sodium ER NDC 0781-8017 by Sandoz Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Structural formula - image 01

The text seems to be a chemical formula and molecular weight. The chemical formula reads CaaHasFNO and is followed by the molecular weight which is 433.46 g/mol.*
Figure-2 - image 03

The text is a table presenting the results of a study comparing the effects of Fluvastatin versus Placebo on recurrent cardiac events. The study had 844 patients in the Fluvastatin group and 833 patients in the Placebo group, and primary endpoint (recurrent cardiac events) occurred in 22.5% of patients in the Fluvastatin group compared to an unspecified percentage in the Placebo group. The table also shows results for secondary endpoints. A note at the bottom of the table states that revascularization procedures of the target lesion within the first 6 months of the initial procedure were excluded.*
Figure-3 - image 04

This appears to be a visual representation of the change in Minimum Lumen Diameter (mm) from a study involving Fluvastatin and Placebo. The graph shows the change in diameter for Monotherapy, All Patients, and Combination treatments, represented by different colored lines. The graph also displays the statistical significance of the findings, expressed as p-values. The study also involved the use of cholestyramine therapy.*
Figure-4 - image 05
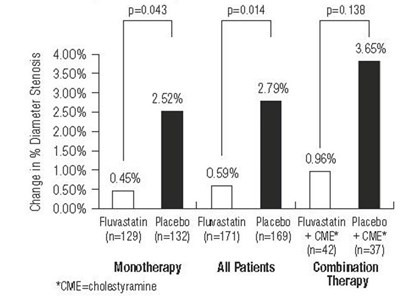
This is a chart showing the change in percentage diameter stenosis. The chart includes data for Fluvastatin, Placebo and Combination therapy, along with the number of patients in each group. The chart also includes the abbreviation CME, which stands for cholestyramine.*
Fluvastatin-xl-100count-Label - image 06

This is a description of a medication with the NDC code 0781-8017-01, which is identified as Fluvastatin Sodium. The medication is in the form of extended-release tablets with a strength of 80mg and is available only with a prescription (RxOnly). The package contains 100 tablets and is manufactured by Sandoz.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
