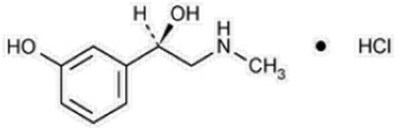
Product Images Phenylephrine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Phenylephrine Hydrochloride NDC 0781-9226 by Sandoz Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1ml-carton - phenyl hcl novaplus 02

Phenylephrine HCl Injection, USP is a medication that needs to be diluted and protected from light. It should be stored in its carton until ready for use. Novaplus 101 ml single-dose vials should have their unused portion discarded. The rest of the text is not available or unreadable.*
5ml-carton - phenyl hcl novaplus 03

Phenylephrine HCI Injection, USP is a Pharmacy Bulk Package that should not be directly infused. It must be diluted and protected from light. It is stored in a carton until the time of use. The package contains 10x 5 mL vials.*
10ml-carton - phenyl hcl novaplus 04

Phenylephrine HCI is a medication that comes in a Pharmacy Bulk Package in a carton. It should be stored in a suitable work area with protection from light until it is time to be used. The medication is not for direct infusion and should be diluted prior to use. Each 10 mL vial contains 10mg of Phenylephrine Hydrochloride along with sodium chloride, sodium citrate dihydrate, citric acid monohydrate, sodium metabisulfite, and water for injection. The pH level is adjusted with Sodium Hydroxide and/or Hydrochloric Acid if necessary. The medication is manufactured in Germany and distributed by Sande b in Princeton, U.S. The package also includes a package insert with directions for use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.