FDA Label for Banophen
View Indications, Usage & Precautions
- OTHER
- ACTIVE INGREDIENT (IN EACH 5 ML)
- PURPOSE
- USES
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- OTC - ASK DOCTOR/PHARMACIST
- WHEN USING THIS PRODUCT
- OTC - PREGNANCY OR BREAST FEEDING
- OTC - KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- PRINCIPAL DISPLAY PANEL - 118 ML BOTTLE LABEL
Banophen Product Label
The following document was submitted to the FDA by the labeler of this product Major Pharmaceuticals. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
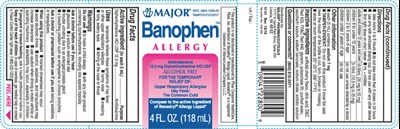
Drug Facts
Distributed by:
MAJOR® PHARMACEUTICALS
17177 N Laurel Park Drive, Suite 233
Livonia, MI 48152
Active Ingredient (In Each 5 Ml)
Diphenhydramine HCl USP 12.5 mg
Purpose
Antihistamine
Uses
temporarily relieves these symptoms of hay fever or other upper respiratory allergies
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Do Not Use
- to make a child sleepy
- with any other product containing diphenhydramine, including one applied topically.
Ask A Doctor Before Use If You Have
- glaucoma
- a sodium restricted diet
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
Otc - Ask Doctor/Pharmacist
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers.
When Using This Product
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Otc - Pregnancy Or Breast Feeding
If pregnant or breast-feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Directions
- take every 4 to 6 hours
- do not take more than 6 doses in 24 hours
- use only with enclosed dosing cup. Do not use with any other device
- find right dose on chart below
- mL = milliliter
| adults and children 12 years and over | 10-20mL (25 mg to 50 mg) |
| children 6 to under 12 years | 5-10mL (12.5 mg to 25 mg) |
| children 2 to 5 years of age | do not use unless directed by a doctor |
| children under 2 years of age | do not use |
Other Information
- each (5 mL) contains: sodium 7 mg
- TAMPER-EVIDENT: Do not use this product if inner foil seal over the mouth of the bottle is cut, torn, broken or missing
- store at room temperature 20°-25°C (68°-77°F)
Inactive Ingredients
artificial cherry flavor, citric acid, D&C Red #33, FD&C Red #40, glycerin, polysorbate 20, purified water, saccharin sodium, sodium benzoate, sodium citrate, sorbitol solution
Questions Or Comments?
(800) 616-2471
Principal Display Panel - 118 Ml Bottle Label
MAJOR®
NDC 0904-1228-00
TAMPER-EVIDENT
Banophen™
ALLERGY
Antihistamine
12.5 mg Diphenhydramine HCl USP
ALCOHOL FREE
FOR THE TEMPORARY
RELIEF OF:
Upper Respiratory Allergies
Hay Fever
The Common Cold
Compare to the active ingredient
of Benadryl® Allergy Liquid*
4 FL. OZ. (118 mL)
* Please review the disclaimer below.