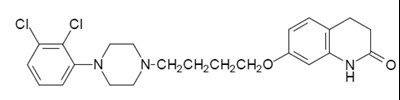Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
- da814597 50e5 48fa e053 2995a90ab21b 00
- image fig1 effectofotherdrug arip
- image fig2 effectofotherdrug dehydroarip
- image fig3 effectofarip otherdrug
- image fig4 effectofintrinsicfactor arip
- image fig5 effectofintrinsicfactor dehydroarip
- image fig6 schizophreniastudy 5
- image fig7 bipolarstudy 7
- image fig8 bipolarstudy 8
- image fig9 tourettesdisorderstudy1
- image logo medguide
- image logo pi
- image structure
Product Label Images
The following 13 images provide visual information about the product associated with Aripiprazole NDC 0904-6656 by Major Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image fig1 effectofotherdrug arip

This is a table titled "Effect of Other Drugs on Aripiprazole PK" which shows the impact of various drugs on the pharmacokinetics of aripiprazole. The drugs are classified based on their ability to inhibit or induce specific enzymes, such as CYP3A4 and CYP2D6, which are involved in aripiprazole metabolism. The table also includes the effect of gastric acid blockers and other drugs like valproate, lithium, and lorazepam. The data is presented as fold change and 90% confidence interval relative to reference without interacting drug.*
image fig2 effectofotherdrug dehydroarip
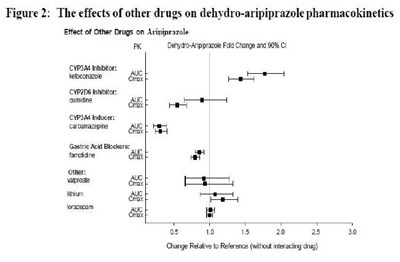
Figure 2 shows the impact of different drugs on the pharmacokinetics of dehydro-aripiprazole. The fold change and 90% confidence interval of the drug's effects are demonstrated. The effects of CYP2D6 and CYP3A4 inhibitors are shown to significantly impact dehydro-aripiprazole metabolism. The chart also shows the effects of gastric acid blockers and risperidone on the drug's metabolism.*
image fig3 effectofarip otherdrug

This is a chart displaying the effects of aripiprazole on the pharmacokinetics of various drugs. It shows the fold change and 50% confidence intervals for drugs such as deropmetoptan, cvpace, artann, omeprazole, lamotigine, valproate, lithium, lorazepam, venidaine, Odesmyhenataing, and escitiopram. The change in relation to the reference without interacting drug is also displayed.*
image fig4 effectofintrinsicfactor arip

The text is a figure caption describing the effects of intrinsic factors on aripiprazole pharmacokinetics, including different populations, such as poor vs. extensive metabolizers of CYP206, males vs. females, and people of different ages and with hepatic or renal impairments. The text provides fold change and 90% CI for each group compared to the reference and graphs of change relative to reference over time.*
image fig5 effectofintrinsicfactor dehydroarip
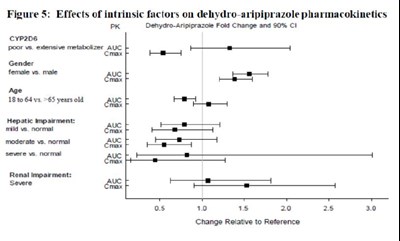
The text provides a figure showing the impact of various intrinsic factors on the pharmacokinetics of dehydro-aripiprazole. The factors included are CYP2D6 metabolism status, gender, age, hepatic impairment severity, and renal impairment severity. The figure shows fold change and S0% CI values for each factor compared to the reference measurement. The data is depicted in a graph with a horizontal axis labeled with percentage values and the various factors listed as vertical bars. The text does not provide any additional information.*
image fig7 bipolarstudy 7

The image shows the Kaplan-Meier estimation of the cumulative proportion of patients with relapse in a bipolar study. The graph displays two lines, one for aripiprazole and the other for placebo. The number of subjects at risk in each group is also shown. The x-axis shows the days from randomization.*
image fig8 bipolarstudy 8

The figure shows the Kaplan-Meier estimation of the cumulative proportion of patients experiencing a relapse to any mood event in Bipolar Study 8. The y-axis represents the proportion with relapse, while the x-axis shows the number of days from randomization. The data includes patients who were treated with ABILIFY and LACE.*
image fig9 tourettesdisorderstudy1
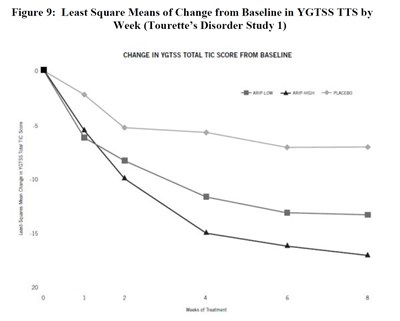
The text appears to be a figure caption displaying the change from baseline in a Tourette's Disorder study. It includes a graph displaying the Least Square Means and Weeks of Treatment for YGTSS TTS. It is not possible to provide a more specific description without seeing the actual figure.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.