Product Images Duloxetine
View Photos of Packaging, Labels & Appearance
- 530e2d92 592c 488e 9a3b 1a61f09e6fc1 00
- 530e2d92 592c 488e 9a3b 1a61f09e6fc1 01
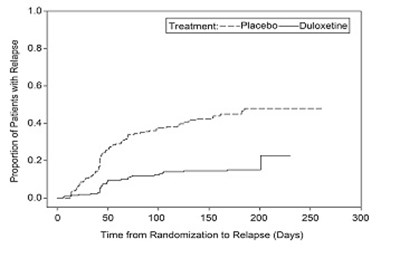
- Figure 1: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (MDD Study 5) - duloxetine fig1
- Figure-2 - duloxetine fig2
- Figure-3 - duloxetine fig3
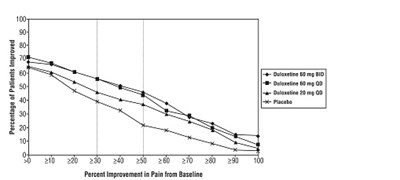
- Figure 4: Percentage of DPNP Adult Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study DPNP-2) - duloxetine fig4
- Figure 5 - duloxetine fig5
- Figure 6 - duloxetine fig6
- Figure 7 - duloxetine fig7
- Figure 8 - duloxetine fig8
- Figure 10: Percentage of Adult Patients with OA Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study OA-1) - duloxetine fig9
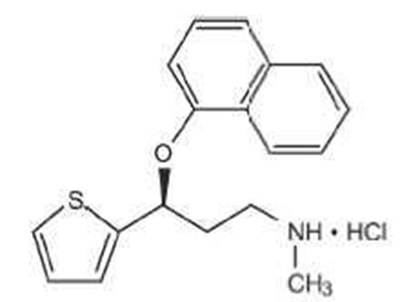
- Chemical Structure - duloxetine str
- carton label - image 02
Product Label Images
The following 13 images provide visual information about the product associated with Duloxetine NDC 0904-7044 by Major Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
530e2d92 592c 488e 9a3b 1a61f09e6fc1 00

This is a description of a medication called Duloxetine delayed-release capsules, which is only available by prescription. The medication comes in a 100-capsule package and is distributed by the company MAJOR. The National Drug Code (NDC) for the medication is 0904-7044-61 and it comes with a medication guide that should be dispensed by the pharmacist. The dosage and other important information about the medication can be found on the package insert.*
530e2d92 592c 488e 9a3b 1a61f09e6fc1 01

This is a description of a medication called Duloxetine. It comes in delayed-release capsules with NDC code 0904-7043-61 and is to be dispensed with a medication guide. It is only available with a prescription and there are 100 capsules per unit dose. The manufacturer is called Major and the address is included as 7N Caur Pk. St 353, Uvonts i 43183 USA.*
Figure 1: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (MDD Study 5) - duloxetine fig1

Figure 4: Percentage of DPNP Adult Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study DPNP-2) - duloxetine fig4

This is a graph showing the percentage of patients who improved in pain when taking Duloxetine (60mg BID, 60mg QD) or Placebo. The X-axis represents the percentage improvement in pain from baseline and the Y-axis represents the percentage of patients improved. The graph shows that more patients improved when taking Duloxetine compared to Placebo.*
Figure 5 - duloxetine fig5

The text describes a graph showing the percentage of patients who have shown improvement in pain and their level of improvement after taking various medications. There are three medication options: Duloxetine 60 mg twice a day, Duloxetine 60 mg once a day, and a placebo. The X-axis shows the percent improvement in pain from baseline (starting point). The Y-axis shows the percentage of patients who have improved. The graph shows that a higher percentage of patients have improved with Duloxetine compared to the placebo. However, more patients have shown improvement with Duloxetine twice daily compared to once daily. The graph also has multiple markings for improvement levels above 10%, with the highest level being over 80%.*
Figure 6 - duloxetine fig6

This appears to be a table displaying the percent of patients who have improved with different doses of duloxetine (20mg, 60mg, and 120mg) compared to a placebo for pain relief. The numbers on the bottom represent the percent improvement in pain from baseline, ranging from 0 to 100.*
Figure 7 - duloxetine fig7

This is a chart showing the percentage of patients improved with either a Placebo or Duloxetine (at doses of 60/120mg) once. The chart also displays percent improvement in pain from Baseline (BOCF).*
Figure 8 - duloxetine fig8

The text provides a graph showing the percentage of patients improved with placebo and duloxetine medication taken once daily. The X-axis indicates the percent improvement in pain from baseline, and the Y-axis indicates the number of patients. However, there is not enough context to determine the nature of the study or the medical condition under consideration.*
Figure 10: Percentage of Adult Patients with OA Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study OA-1) - duloxetine fig9

carton label - image 02

This is a description of Duloxetine delayed-release capsules USP manufactured by Major Pharmaceuticals. The NDC number is 0904-7045-61, and each unit dose contains 100 capsules. These capsules should be dispensed by a pharmacist with a medication guide. The dosage is 30-60mg per day, depending on the patient's condition. The medication is indicated for Major Depressive Disorder (MDD), Generalized Anxiety Disorder (GAD), Diabetic Peripheral Neuropathic Pain (DPNP), and Fibromyalgia (FM). The text also includes instructions for storage, and a lot number and expiration date for the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.