Product Images Recombinate
View Photos of Packaging, Labels & Appearance
Product Label Images
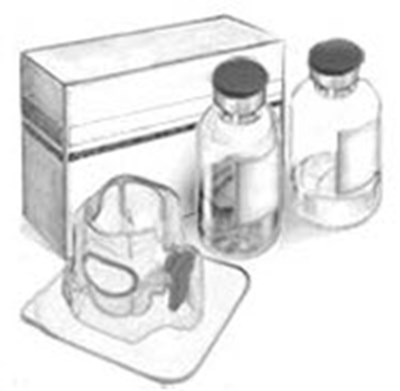
The following 25 images provide visual information about the product associated with Recombinate NDC 0944-2841 by Takeda Pharmaceuticals America, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
recombinate 15

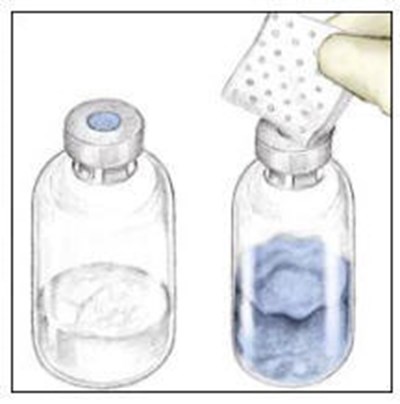
This appears to be a product label for a sterile water for injection solution that is intended to be used to reconstitute another product. The solution comes in a single-dose container, and cautionary instructions are provided such as the need to ensure the solution is clear and the recommendation to add a suitable solute to make it isotonic if used for intravascular injection. The product is nonpyrogenic.*
recombinate 16

RECOMBINATE is an antihemophilic factor used for intravenous administration only. It should be stored in a refrigerator at 36° to 46°F (2° to 8°C) or at room temperature up to 86°F (30°C). Once reconstituted, it should not be refrigerated. The medication does not contain any preservatives. The package insert should be read before using this medication. It is manufactured by Baxalta US Inc. with the U.S. License No. 2020 RcOnly. Lot number, expiration date, date of the dose, and time of the dose fields are available to be filled out on the packaging.*
recombinate 17
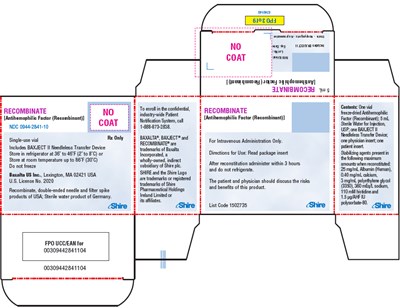
This appears to be a description of a medical product named "RECOMBINATE". The text includes information on how to use and store the product, including directions for use and storage temperatures. The product seems to be related to treating hemophilia and has a confidential patient registration system in place. The text also includes a company name "Baxter Healthcare Corporation" and a license number "2020" related to the product. However, there is insufficient information to provide a full description of the product's benefits and application.*
recombinate 18

RECOMBINATE is an antihemophilic factor (recombinant) indicated for intravenous administration only. It should be stored in a refrigerator at 36° to 46°F (2° to 8°C) or at room temperature up to 86°F (30°C). Reconstituted RECOMBINATE should not be refrigerated and contains no preservative. The direction for use is available in the package insert. This product is produced and distributed by Baxalta US Inc. The lot and expiration dates are specified in the text.*
recombinate 19
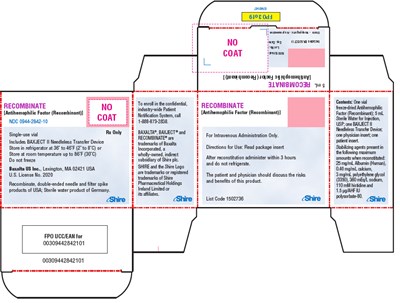
This is a description of a medical product with the text obtained by . The text contains information about the product's contents, storage conditions, and usage directions, along with a trademark and license number of the manufacturer. The product is indicated for administration with a double-ended needle and a spike, and it is recommended to store the product in a refrigerator or at room temperature, depending on the temperature range. The risks and benefits of the product should be discussed between patients and physicians.*
recombinate 20

This appears to be a label for a vial of RECOMBINATE, a medication used for intravenous administration to treat bleeding disorders caused by low levels of clotting Factor VIII. The label provides information on the lot number, expiration date, and storage instructions. There is also a mention of a package insert with directions for use.*
recombinate 21
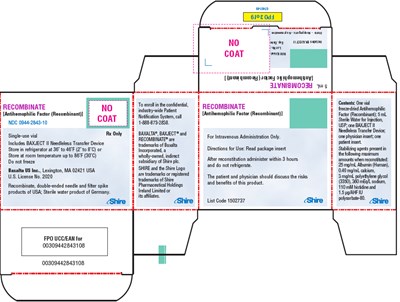
This text is a product label for Recombinate, an Antihemophilic Factor (Recombinant) used for treating Hemophilia A. It contains information about the storage of the product, directions for use, and possible side effects. It also includes a unique product code (0030942843108) and the manufacturer's information.*
recombinate 22

RECOMBINATE is a medication used for intravenous administration to treat hemophilia A and B. It is stored in refrigeration at 36 to 46°F (2°t0 8°C) or at room temperature up to 86°F (30°C). After reconstitution, it should not be refrigerated. The medication does not contain preservatives and should be used with caution. The medication's packaging contains the required instructions for use.*
recombinate 23
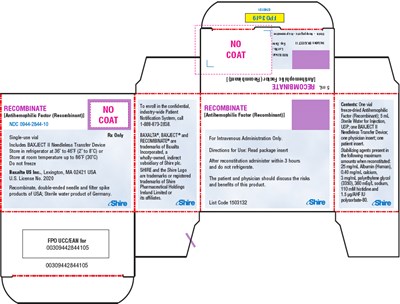
This is a description of a medical product called "Recombinant," mainly used for antihemophilic factor deficiency. It comes with a double-ended needle and fitter spike, and it must be refrigerated between 38 to 46 degrees Fahrenheit. The package contains instructions for use and a patient notification system. The list of contents includes Oz il, and it is manufactured by Baxalta US Inc. Any patient, before using this product, must discuss with their physician the possible benefits and side effects, such as calcium, sodium, or histidine. The numerical codes 6196 0dd and 0030942844105 are also present.*
recombinate 24

This is a product label for Recombinate, an antihemophilic factor (recombinant) used for intravenous administration. It includes a lot number, expiration date, and instructions for proper storage both before and after reconstitution. There are also warnings about not refrigerating after reconstitution and directions to read the package insert. The label is produced by Baxalta US Inc. and includes a trademark symbol.*
recombinate 25
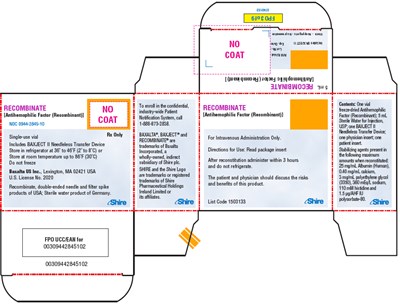
This appears to be a description of a medication, likely for a form of hemophilia or blood clotting disorder. The text includes instructions for use and storage, as well as a phone number to enroll in a confidential patient notification system. However, much of the text is garbled and non-readable due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.