Product Images Advate
View Photos of Packaging, Labels & Appearance
- Figure A - advate 01
- Figure B - advate 02
- Figure C - advate 03
- Figure D - advate 04
- Figure E - advate 05
- Image - advate 06
- Image - advate 07
- Image - advate 08
- Image - advate 09
- Image - advate 10
- Image - advate 11
- Image - advate 12
- Figure - advate 13
- Figure - advate 14
- Figure - advate 15
- Figure - advate 16
- Figure - advate 17
- Figure - advate 18
- Figure - advate 19
- advate 20
- advate 21
- advate 22
- advate 23
- advate 24
- advate 25
- advate 26
- advate 27
- advate 28
- advate 29
- advate 30
- advate 31
- advate 32
- advate 33
- advate 34
- advate 35
- advate 36
- advate 37
- advate 38
- advate 39
- advate 40
- advate 41
- advate 42
Product Label Images
The following 42 images provide visual information about the product associated with Advate NDC 0944-3047 by Takeda Pharmaceuticals America, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - advate 06

This is a color descriptor for a shade of blue that is pale or pastel in nature, often associated with a light and airy feeling.*
Image - advate 12

This text is a color name representation and does not provide enough context to generate a useful description.*
advate 21

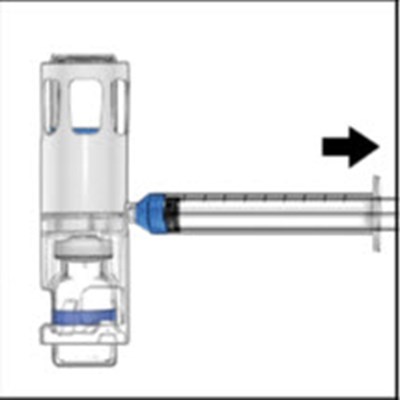
ADVATE is a recombinant Antihemophilic Factor used for intravenous administration only. It is a lyophilized powder that requires reconstitution using the diluent preassembled in the BAXJECT II system. It is a single-use product and does not contain plasma or albumen. ADVATE has trademarks of Shire Pharmaceutical and should be stored at a temperature of 2°-6° and should not be refrigerated after reconstitution. Administration should be done within 3 hours of reconstitution.*
advate 23

This appears to be a product description of a medical item called ADVATE, which is used for intravenous administration. The product does not contain plasma/albumin and has a unique identifier of 0944-3052-03. The package is single-use only, and caution is advised to not open the packaging until time of use. The rest of the text seems to be product codes and identifiers, including a RSS 14 stacked barcode.*
advate 24

ADVATE is a powder for reconstitution with the diluent preassembled in BAXJECT II, which is used for antihemophilic factor (recombinant) treatment. It is for single use only, does not contain plasma/albumin and should be administered within 8 hours of reconstitution. ADVATE should be stored between 2-8°C (up to 85°F) and not be used after 24 months. This medication should not be frozen or refrigerated after reconstitution. For more information about ADVATE, visit the website advate.com.*
advate 26

The text describes a medical product called Antihemophilic Factor (Recombinant), administered intravenously, with a single-use only tag. It is advised to use the product before the mentioned time of use and discard in case of open or damaged packaging.*
advate 27

ADVATE is a powdered medication used for intravenous administration only. It is a recombinant antihemophilic factor and does not contain plasma or albumin. The product comes preassembled with a diluent in BAXJECT Il syringe, and it is for single use only. Additional information on storage and administration details are mentioned on the label.*
advate 28

This is a description of a medical product called ADVATE. It is a recombinant antihemophilic factor that should be administered intravenously only. The product comes preassembled with diluent in a BAXJECT II system. It does not contain plasma or albumin. The text also includes a barcode number and a reference to Shire, which may be the manufacturer of the product.*
advate 29

This appears to be a description of a medical product called "ADVATE 3", which is an intravenous medication used to treat hemophilia. The product comes preassembled in packaging and should only be used once. Additionally, it does not contain plasma or albumin.*
advate 30

ADVATE is an antihemophilic factor which is recombinant in nature. It is available as a powdered form that can be reconstituted with diluent preassembled in BAXJECT II. This product does not contain any plasma albumin. The product is meant for single use only. The potency of ADVATE has been mentioned in the text. The storage temperature range for the product is also given. The product cannot be used beyond its expiry date. The instructions for administration have also been given in the text. The website for ADVATE is mentioned at the end.*
advate 31

The text is a product label for ADVATE, which is a recombinant antihemophilic factor used for intravenous administration. The medication is preassembled in a BAXJECT II syringe and does not contain plasma/albumin. Instructions for use can be found in the package insert.*
advate 32

The text describes a medical product called "Antihemophilic Factor (Recombinant)" which is for intravenous administration only and is preassembled in the BAXJECT Ill system. It should only be used once and if the packaging is opened or damaged, it should not be used. The product does not contain plasma/albumin. The rest of the text is not legible.*
advate 33

ADVATE is a lyophilized powder for intravenous administration, containing antihemophilic factor (recombinant). The product should be reconstituted using the BAXJECT I system and is for single use only, as it does not contain plasma or albumin. Storage temperature ranges from 2-8°C or up to 5°F (30°) for a period of up to 6 months. The product contains stabilizing agents and should be administered within 3 hours of reconstitution and should not be subjected to agitation post-reconstitution. The manufacturer, Shire, provides a package insert that outlines the benefits and risks associated with ADVATE.*
advate 34

ADVATE is a recombinant antihemophilic factor designed for intravenous administration. The product is preassembled with diluent in a BAXJECT Ill system. To learn how to use the product, refer to the package insert. ADVATE does not contain plasma or albumin. The rest of the text appears to be product codes or serial numbers and is not relevant to the description.*
advate 35

This is a description of a medical product called ADVATE, which is a recombinant Antlhemnphlllc Factor. The packaging contains a preassembled ADVATE and diluent. The product does not contain plasma/albumin. It must be administered intravenously and is for single use only. The packaging should not be opened or used if damaged. The LOT number is 3.*
advate 36

ADVATE is a lyophilized powder for reconstitution containing Antihemophilic Factor (Recombinant) and is administered intravenously. It comes with diluent preassembled in BAXJECT Il system and does not contain plasma/albunin. ADVATE is a product of Bt Inceporte and has stabilizing agents present in maximum amounts when reconstituted. The product should be stored at 36° 0 46°F (2°-8°) or room temperature (up to 85°F [30°]) for a period of up to 6 months. It should not be used beyond expiration date and should be administered within 3 hours of reconstitution and not refrigerated after reconstitution. The package contains 5 vials of Antihemophilic Factor (Recombinant) and a package insert. ADVATE is a product of Swnd and Shire is a registered trademark of S Pharmaceutical Holdings.*
advate 37

ADVATE is a medication used for intravenous administration to treat Antihemophilic factor (Recombinant) deficiency. The medication and diluent are preassembled in the BAXJECT III system. The package insert should be consulted for directions for use. The product does not contain plasma or albumin. Additional information is not-available.*
advate 38

This is a description for a medical product called "Antihemophilic Factor (Recombinant)", which comes in a preassembled set with a diluent and should not be opened until it's time for use. It's specified that it's for intravenous administration only and that if the packaging is opened or damaged it shouldn't be used. The product doesn't contain plasma/albumin. There is also a code, possibly for tracking or inventory purposes, shown as "0030944304716".*
advate 39

ADVATE is a medication that is given intravenously. It is a lyophilized powder that needs to be mixed to be used. It comes preassembled with diluent in the BAXJECT 1 system. ADVATE does not contain plasma or albumin. Some stabilizing agents exist in it during reconstitution in specific amounts. ADVATE is a trade name of Shire Pharmaceuticals Holding LLC, which is a wholly-owned subsidiary of Shire Inc. It should be stored between 36 and 46 degrees Fahrenheit. ADVATE has an expiration date and should not be used beyond that date. It is important to discuss the drug's risks and benefits with a doctor before use. ADVATE is a product of Switzerland and is processed in Germany or the United States using the BAXJECT I system.*
advate 40

ADVATE is a recombinant antihemophilic factor for intravenous administration only. The product is preassembled with diluent in the BAXJECT III system. Directions for use are available in the package insert. The product does not contain plasma or albumin. The rest of the text is not available or not readable.*
advate 41

Antihemophilic Factor (Recombinant) is a medication produced by Shire with the reference code 0742475. Further information is not available.*
advate 42

This is a product label or packaging label for Sterile Water for Injection manufactured by Siegfried Hameln GmbH. The lot number for this particular batch is not provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.