Product Images Adynovate
View Photos of Packaging, Labels & Appearance
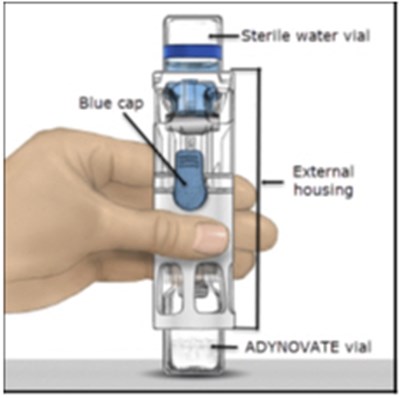
- Figure A - adynovate 01
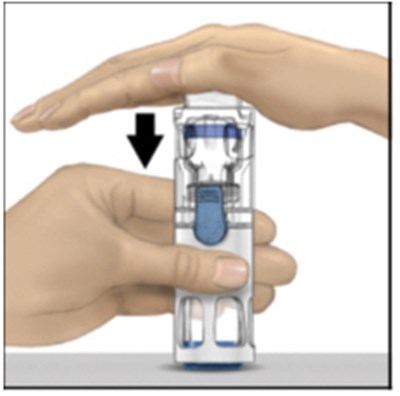
- Figure B - adynovate 02
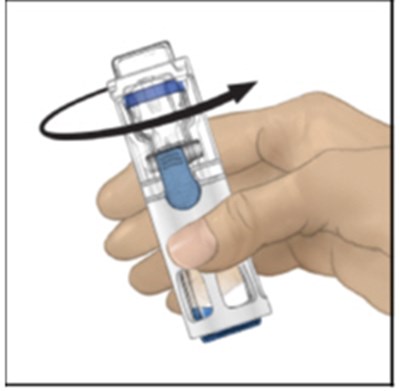
- Figure C - adynovate 03
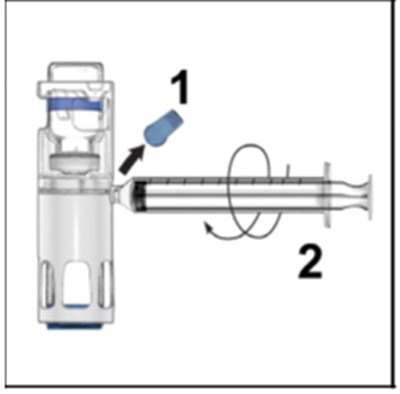
- Figure D - adynovate 04
- Figure E - adynovate 05
- image - adynovate 06
- image - adynovate 07
- image - adynovate 08
- image - adynovate 09
- image - adynovate 10
- image - adynovate 11
- image - adynovate 12
- image - adynovate 13
- image - adynovate 14
- image - adynovate 15
- image - adynovate 16
- image - adynovate 17
- image - adynovate 18
- image - adynovate 19
- PRINCIPAL DISPLAY PANEL - Kit Carton - 250IU - adynovate 20
- PRINCIPAL DISPLAY PANEL - Barrel Label - 250IU - adynovate 21
- PRINCIPAL DISPLAY PANEL - Blister Label - 250IU - adynovate 22
- PRINCIPAL DISPLAY PANEL - Kit Carton - 500IU - adynovate 23
- PRINCIPAL DISPLAY PANEL - Barrel Label - 500IU - adynovate 24
- PRINCIPAL DISPLAY PANEL - Blister Label - 500IU - adynovate 25
- PRINCIPAL DISPLAY PANEL - Kit Carton - 750IU - adynovate 26
- PRINCIPAL DISPLAY PANEL - Barrel Label - 750IU - adynovate 27
- PRINCIPAL DISPLAY PANEL - Blister Label - 750IU - adynovate 28
- PRINCIPAL DISPLAY PANEL - Kit Carton - 1000IU - adynovate 29
- PRINCIPAL DISPLAY PANEL - Barrel Label - 1000IU - adynovate 30
- PRINCIPAL DISPLAY PANEL - Blister Label - 1000IU - adynovate 31
- PRINCIPAL DISPLAY PANEL - Kit Carton - 1500IU - adynovate 32
- PRINCIPAL DISPLAY PANEL - Barrel Label - 1500IU - adynovate 33
- PRINCIPAL DISPLAY PANEL - Blister Label - 1500IU - adynovate 34
- PRINCIPAL DISPLAY PANEL - Kit Carton - 2000IU - adynovate 35
- PRINCIPAL DISPLAY PANEL - Barrel Label - 2000IU - adynovate 36
- PRINCIPAL DISPLAY PANEL - Blister Label - 2000IU - adynovate 37
- PRINCIPAL DISPLAY PANEL - Kit Carton - 3000IU - adynovate 38
- PRINCIPAL DISPLAY PANEL - Barrel Label - 3000IU - adynovate 39
- PRINCIPAL DISPLAY PANEL - Blister Label - 3000IU - adynovate 40
- PRINCIPAL DISPLAY PANEL - Antihemophilic Factor Vial Label - adynovate 41
- PRINCIPAL DISPLAY PANEL - Sterile Water for Injection Vial Label - adynovate 42
Product Label Images
The following 42 images provide visual information about the product associated with Adynovate NDC 0944-4626 by Takeda Pharmaceuticals Amercia, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image - adynovate 06

There is not much context to evaluate. "Light Blue" seems to be a color, and the text appears to be readable. Therefore, the description is: "Light Blue is a color."*
image - adynovate 09

This is a color description and indicates a shade of green that is light in hue.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 250IU - adynovate 20

ADYNOVATE is a medication for intravenous administration used as a treatment for hemophilia. It contains Recombinant PEGylated Anthemophiic Factor and should only be used for intravenous administration. Specific instructions for administration and preparation are not available from the text provided.*
PRINCIPAL DISPLAY PANEL - Barrel Label - 250IU - adynovate 21

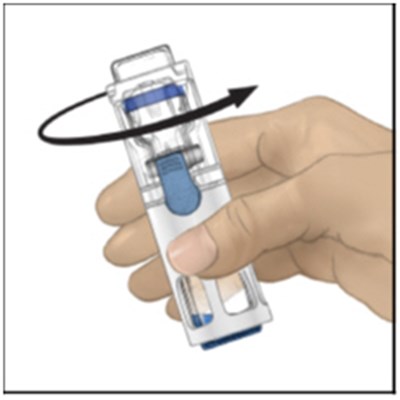
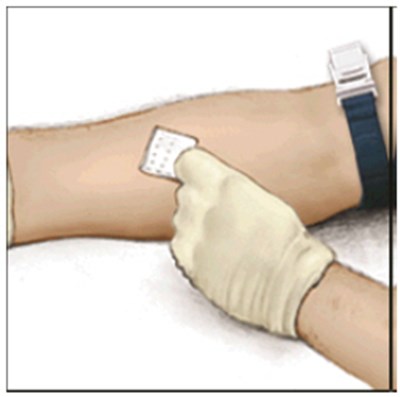
This appears to be a label or instruction manual for the medical product ADYNOVATE, which is administered intravenously using a preassembled BAXJECT IIl system. The directions for use are provided in the package insert. The label also includes a warning that the product is for prescription use only. No further information is available from this text.*
PRINCIPAL DISPLAY PANEL - Blister Label - 250IU - adynovate 22

ADYNOVATE is an antihemophilic factor that is administered intravenously. It is PEGylated and should be used immediately once the packaging is opened. The product comes in a single-use package and if it is damaged, it should not be used. The text also includes a product code (NOC 0944-4622-02).*
PRINCIPAL DISPLAY PANEL - Kit Carton - 500IU - adynovate 23
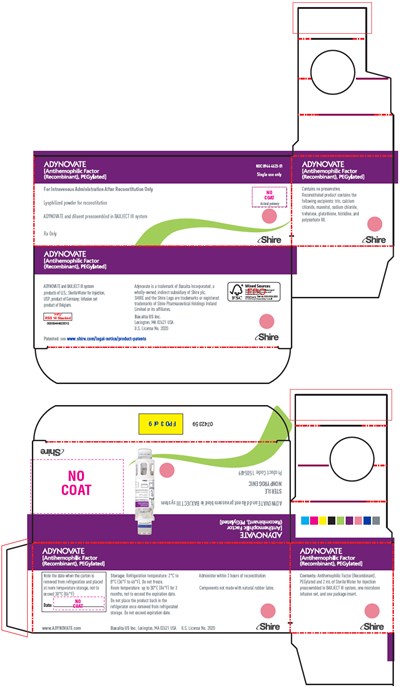
ADYNOVATE is a medication used for intravenous administration after reconstitution. It is a powdered medicine with a potency of 1.8 and is pegylated. It is preassembled in a syringe. The storage requirements dictate storage at room temperature, and the medication must be used promptly after opening. The medication packaging has the Shire logo. The information in the text is limited, and it is advisable to seek additional resources for complete information.*
PRINCIPAL DISPLAY PANEL - Barrel Label - 500IU - adynovate 24

ADYNOVATE is a medication for intravenous administration that contains recombinant antihemophilic factor that has been PEGylated. It comes preassembled with diluent in BAXJECT III system. For more information on how to use ADYNOVATE, please refer to the package insert. This medication requires a prescription. Non-available.*
PRINCIPAL DISPLAY PANEL - Blister Label - 500IU - adynovate 25

This is a description of a drug called "ADYNOVATE" that is administered intravenously. It is an Antihemophilic Factor that is PEGylated and should only be used with the ADINOVATE and st rassembled n BAUECT I sytem. The packaging should not be used if opened or damaged. The given code is NOC 0944-4623-02 and it should be used for single use only.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 750IU - adynovate 26

ADYNOVATE is a recombinant antihemophilic factor that is polyethylene glycolated. It is available in a lyophilized powder form for intravenous administration only. ADYNOVATE comes preassembled in ADVATE or BAXJECT II systems. The product must be stored in refrigerated conditions and once reconstituted, it can be stored at room temperature for up to 3 hours. The package insert provides detailed information on dosage, administration, precautions, and adverse reactions.*
PRINCIPAL DISPLAY PANEL - Barrel Label - 750IU - adynovate 27

This appears to be information regarding the administration of a medication called ADYNOVATE. It is a recombinant medication that has been PEGylated, and is meant for intravenous use only. The medication and its diluent come preassembled in a system called BAXJECT IIl. There is a reference to a package insert for directions on how to use the medication. The other numbers and text do not provide any additional relevant information.*
PRINCIPAL DISPLAY PANEL - Blister Label - 750IU - adynovate 28

Description: "ADYNOVATE" is an antihemophilic factor that is administered intravenously. The product comes in a stacked packaging design, and should not be used if the packaging is opened or damaged. The item is for single use only.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 1000IU - adynovate 29

ADYNOVATE is a recombinant, PEGylated antihemophilic factor used for intravenous administration after reconstitution. The drug comes in the form of a lyophilized powder for reconstitution and is only for administration by a healthcare professional. ADYNOVATE is available in a preassembled BALECT 1 system. The storage requirement is refrigerated, and the drug should not be shaken after reconstitution. The packaging contains 2 mL of solution, and the drug is used to treat bleeding tendencies in people with hemophilia A.*
PRINCIPAL DISPLAY PANEL - Barrel Label - 1000IU - adynovate 30

ADYNOVATE is a prescription drug that is intended for intravenous administration only. It comes preassembled with diluent in the BAXJECT II system. The package insert contains the directions for use. Unfortunately, no further information can be extracted due to the limited and fragmented text.*
PRINCIPAL DISPLAY PANEL - Blister Label - 1000IU - adynovate 31

ADYNOVATE is a recombinant antihemophilic factor that is PEGylated and is administered intravenously. It is provided in a stacked Rss 1 configuration and should not be used if the packaging is opened or damaged. The product uses the BAUECT I system and is for single use only. NOC 0944-4624-02 is the product code.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 1500IU - adynovate 32

ADYNOVATE is a medication used for intravenous administration after reconstitution. It is a recombinant antihemophilic factor that has been PEGylated. The package contains a lyophilized powder that needs to be reconstituted. ADYNOVATE is preassembled in a BALECT1 system. This product needs to be stored at room temperature. The medication is produced by Shire and is available in the UK. The text also contains information about storage, packaging, and the Shire logo.*
PRINCIPAL DISPLAY PANEL - Barrel Label - 1500IU - adynovate 33

This is a package insert of ADYNOVATE, a drug that is for intravenous administration only. The drug and its diluent come preassembled in the BAXJECT III system, and the package contains directions for use. The rest of the text appears to be product codes and may not be useful for generating a description.*
PRINCIPAL DISPLAY PANEL - Blister Label - 1500IU - adynovate 34

Description: ADYNOVATE is an antihemophilic factor used for intravenous administration. It is a recombinant factor that has been PEGylated. It is important to note that ADYNOVATE is only for single use and must not be used if the packaging is damaged or opened.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 2000IU - adynovate 35
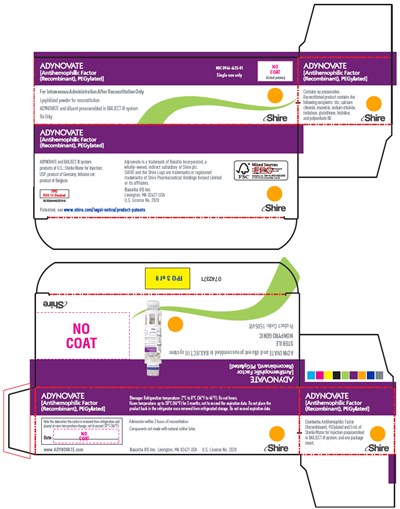
ADYNOVATE is a medication used for the treatment of Hemophilia A, a rare genetic blood clotting disorder caused by the deficiency of clotting factor VIII. It is a Recombinant PEGylated Antihemophilic factor that can only be administered after reconstitution. The medication is presented in a blue color coded vial with a S logo and an identification number. The storage temperature should be between 2-8°C. The package has information on the medication's composition, usage, and storage recommendations, as well as the manufacturer's contact information.*
PRINCIPAL DISPLAY PANEL - Blister Label - 2000IU - adynovate 37

ADYNOVATE is a recombinant antihemophilic factor that is PEGylated for intravenous administration. It is provided in a preassembled system and should only be used once. Users are advised not to use if packaging is opened or damaged. The product's NDC number is 0944-4625-02 and it is for single use only.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 3000IU - adynovate 38

This is a description of a medical product called "ADYNOVATE" indicated for the treatment of hemophilia A. It contains antihemophilic factor (recombinant) PEGylated. The text provides information on the storage temperature and instructions for administration. However, some words are not readable due to errors.*
PRINCIPAL DISPLAY PANEL - Blister Label - 3000IU - adynovate 40

Adynovate 2 is a medication used for intravenous administration to treat hemophilia. This is a pegylated recombinant antihemophilic factor that is used only for single-use. It comes with a preassembled Baxject System and is not stackable. Additional information is not available.*
PRINCIPAL DISPLAY PANEL - Antihemophilic Factor Vial Label - adynovate 41

Antihemophilic Factor (Recombinant), PEGylated is a medication produced by Shire. Its purpose is not clear from the provided text.*
PRINCIPAL DISPLAY PANEL - Sterile Water for Injection Vial Label - adynovate 42

This is a product description for "Sterile Water for Injection" manufactured by Siegfried Hameln GmbH with a product code of 0742211.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.