Product Images Insulin Glargine U-300 Max
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 51 images provide visual information about the product associated with Insulin Glargine U-300 Max NDC 0955-2900 by Winthrop U.s, A Business Of Sanofi-aventis U.s. Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Chemical Structure - insulin glargine 01

This is a graph that shows the Glucose Infusion Rate (GIR) in mg/(kg*min) over time in hours after a subcutaneous injection. The values are presented on a table and a graph but no additional information is available.*
Image - insulin glargine 02

This text provides a list of body parts: Upper arms, stomach and thighs.*
Image - insulin glargine 03

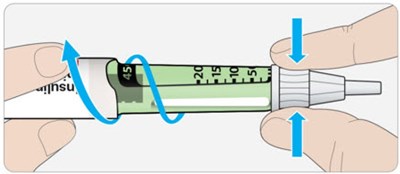
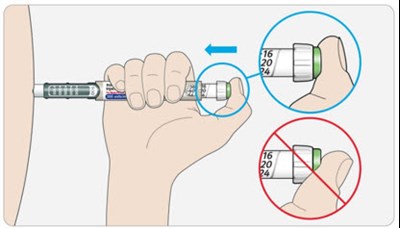
This appears to be a product manual for a device used for administering doses of insulin. The device features a dose window, dose pointer, dose selector, insulin scale, and an injection button. There is also a pen cap and rubber seal. The manual indicates that the plunger will not be visible until after several doses have been administered.*
Image - insulin glargine 26
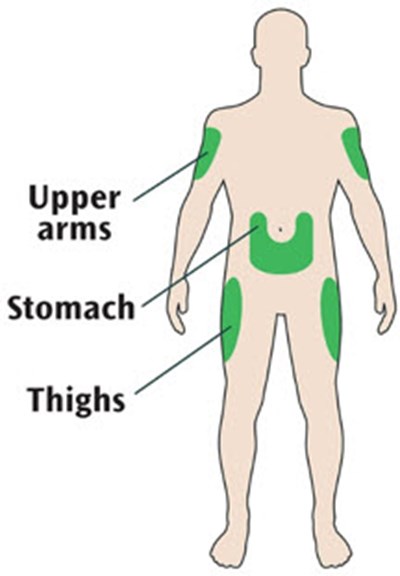
This is a list of body parts that includes the upper arms, stomach and thighs.*
Image - insulin glargine 27

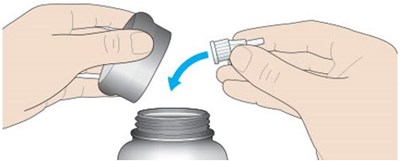
The text describes the parts of a cartridge holder used for injecting insulin. It includes a dose window and pointer, dose selector, injection button, pen cap, rubber seal, insulin scale, and insulin name. The plunger is not visible until after some doses have been administered.*
Image - insulin glargine 31

This appears to be a dosage strength specification for a medication, likely intended for subcutaneous injection. The unit "units/mL" is commonly used to measure the concentration of medications such as insulin. Without further context or information about the specific medication, it is difficult to provide more details.*
PRINCIPAL DISPLAY PANEL - 1.5 mL Pen Carton - insulin glargine 50

This is a description of insulin glargine U-300 SoloStar, which is a disposable insulin delivery device intended for subcutaneous use only. The solution is to be used only for single patient use and should not be removed with a syringe. It is advised to mix it gently before use and only use it if clear and colorless with no visible particles. The prefilled pens are to be used within 56 days after opening and needles are not included. The product is manufactured by Winthrop. GTIN number 0030955390035 is also provided.*
PRINCIPAL DISPLAY PANEL - 3 mL Pen Carton - insulin glargine 51

This is a product description and instructions for use of a disposable insulin delivery device called Insulin glargine U-300 Max SoloStar®. The product is Rx only and is only for single patient use. Two 3mL pre-filled pens are dispensed in a sealed carton and needles are not included. The solution should be clear and colorless with no visible particles and should be used within 28 days of opening. The product should not be mixed with other insulins and always use a new needle for administration. The text also includes some product identification numbers.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.