Product Images Niacin
View Photos of Packaging, Labels & Appearance
- The structural formula of Niacin extended-release tablets are unscored, film-coated tablets for oral administration and are available in two tablet strengths containing 500 mg and 1000 mg niacin. - niacin extended release 01
- PRINCIPAL DISPLAY PANELNDC 10135-0612-90MarlexNiacinExtended- Release Tablets500 mgRx Only90 Tablets - niacin extended release 02
- PRINCIPAL DISPLAY PANELNDC 10135-0613-90MarlexNiacinExtended- Release Tablets750 mgRx Only90 Tablets - niacin extended release 03
- PRINCIPAL DISPLAY PANELNDC 10135-0614-90MarlexNiacinExtended- Release Tablets1000 mgRx Only90 Tablets - niacin extended release 04
Product Label Images
The following 4 images provide visual information about the product associated with Niacin NDC 10135-612 by Marlex Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The structural formula of Niacin extended-release tablets are unscored, film-coated tablets for oral administration and are available in two tablet strengths containing 500 mg and 1000 mg niacin. - niacin extended release 01
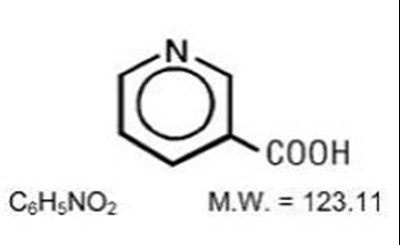
This appears to be a chemical composition description. The compound has a carboxylic acid (COOH) group and a molecular weight (MW) of 123.11 g/mol. The other characters (CsHsNO) may represent the chemical formula of a functional group within the compound, but without more information it is difficult to determine its exact identity.*
PRINCIPAL DISPLAY PANELNDC 10135-0612-90MarlexNiacinExtended- Release Tablets500 mgRx Only90 Tablets - niacin extended release 02
This text is a medication label that provides instructions for pharmacists on how to dispense Niacin Extended-Release Tablets. It includes information on the usual dosage, storage, and container requirements, as well as details on the tablet contents, packaging, and distribution. It also warns against accepting tablets with a broken seal over the bottle opening.*
PRINCIPAL DISPLAY PANELNDC 10135-0613-90MarlexNiacinExtended- Release Tablets750 mgRx Only90 Tablets - niacin extended release 03
This is a medication information sheet for Niacin Extended-Release Tablets, manufactured by Marlex Pharmaceuticals. It contains instructions for dispensing, storage, and dosing, as well as the medication's ingredients and the manufacturer's contact information. The sheet requests that the pharmacist not attach patient information to the package insert. It also specifies that the medication should be dispensed in a container with a child-resistant closure and should not be accepted if the bottle seal is broken or missing. Lot number and expiration date is also specified.*
PRINCIPAL DISPLAY PANELNDC 10135-0614-90MarlexNiacinExtended- Release Tablets1000 mgRx Only90 Tablets - niacin extended release 04
This is a prescription medication for Niacin extended-release tablets. The usual dosage information can be found in the package insert. The pharmacist is instructed to dispense the medication in a tight container with a child-resistant closure and to not accept if the seal is broken or missing. The medication should be stored at room temperature. The medication package contains 90 tablets and has an NDC number of 10135-0614-90. The tablets are manufactured and distributed by Marlex Pharmaceuticals. Lot number and expiration date information is also provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.