Product Images Atorvastatin Calcium
View Photos of Packaging, Labels & Appearance
- Table 3. Clinical adverse reactions occurring in ≥ 2% in patients treated with any dose of atorvastatin calcium and at an incidence greater than placebo regardless of causality (% of patients). - atorvastatin tablet 01
- The structural formula for phenylamino carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1). The empirical formula of atorvastatin calcium is C66H68CaF2N4O10 and its molecular weight is 1155.36. - atorvastatin tablet 02
- TABLE 4. Effect of Co-administered Drugs on the Pharmacokinetics of Atorvastatin - atorvastatin tablet 03
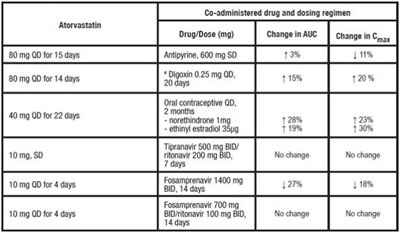
- TABLE 5. Effect of Atorvastatin on the Pharmacokinetics of Co-administered Drugs - atorvastatin tablet 04
- Figure 1: Effect of Atorvastatin Calcium 10 mg/day on Cumulative Incidence of Non Fatal Myocardial Infarction or Coronary Heart Dis ease Death (in ASCOT-LLA) - atorvastatin tablet 05
- Figure 2: Effect of Atorvastatin Calcium 10 mg/day on Time to Occurrence of Major Cardiovascular Event (myocardialinfarction, acute CHD death, unstable angina, coronary revascularization, or stroke) in CARDS - atorvastatin tablet 06
- Figure 3: Effect of Atorvastatin Calcium 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT) - atorvastatin tablet 07
- TABLE 6. Overview of Efficacy Results in TNT - atorvastatin tablet 08
- TABLE 7. Dose Response in Patients With Primary Hyperlipidemia (Adjusted Mean % Change From Baseline)a - atorvastatin tablet 09
- TABLE 8. Mean Percentage Change From Baseline at Endpoint (Double-Blind, Randomized, Active-Controlled Trials) - atorvastatin tablet 0a
- TABLE 9. Combined Patients with Isolated Elevated TG: Median (min, max) Percentage Change From Baseline - atorvastatin tablet 0b
- TABLE 10. Open-Label Cross over Study of 16 Patients With Dysbetalipoproteinemia (Fredricks on Type III) - atorvastatin tablet 0c
- TABLE 11. Lipid-altering Effects of Atorvastatin Calcium in Adolescent Boys and Girls with Heterozygous Familial Hypercholesterolemia or Severe Hypercholesterolemia (Mean Percentage Change From Baseline at Endpoint in Intention-to-Treat Population) - atorvastatin tablet 0d
- PRINCIPAL DISPLAY PANELNDC 10135-0649-10Atorvastatin Calcium Tablets10 mg1000 TABLETSRx Only - atorvastatin tablet 0e
- PRINCIPAL DISPLAY PANELNDC 10135-0650-05Atorvastatin Calcium Tablets20 mg500 TABLETSRx Only - atorvastatin tablet 0f
- PRINCIPAL DISPLAY PANELNDC 10135-0651-10Atorvastatin Calcium Tablets40 mg1000 TABLETSRx Only - atorvastatin tablet 10
- atorvastatin tablet 11
Product Label Images
The following 17 images provide visual information about the product associated with Atorvastatin Calcium NDC 10135-653 by Marlex Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Table 3. Clinical adverse reactions occurring in ≥ 2% in patients treated with any dose of atorvastatin calcium and at an incidence greater than placebo regardless of causality (% of patients). - atorvastatin tablet 01
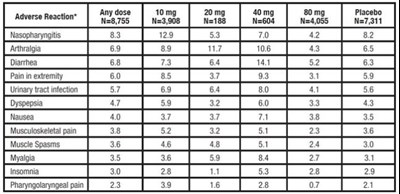
This appears to be a table displaying side effects of a medication called Advers Resction. The left column lists the side effect and the right column lists the percentage of individuals who experienced that side effect at two different doses. Some of the side effects listed include nasopharyngitis, arthralgia, diarrhea, pain in extremity, nausea, musculoskeletal pain, muscle spasms, myalgia, insomnia, and pharyngolaryngeal pain.*
The structural formula for phenylamino carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1). The empirical formula of atorvastatin calcium is C66H68CaF2N4O10 and its molecular weight is 1155.36. - atorvastatin tablet 02
![atorvastatin-tablet-02.jpg The structural formula for phenylamino carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1). The empirical formula of atorvastatin calcium is C66H68CaF2N4O10 and its molecular weight is 1155.36. - atorvastatin tablet 02](https://ndclist.com/assets/spl/images/64abfa40-e0ab-46ae-8c7c-7c199b89b7c1/400x-atorvastatin-tablet-02.jpg)
TABLE 4. Effect of Co-administered Drugs on the Pharmacokinetics of Atorvastatin - atorvastatin tablet 03
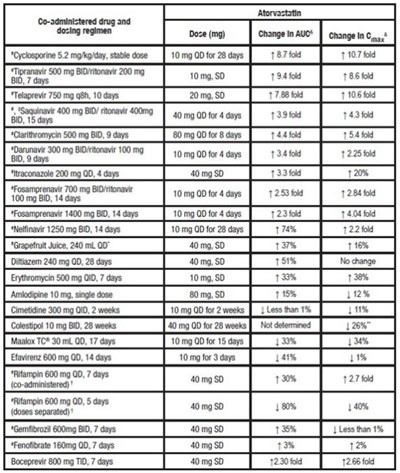
TABLE 5. Effect of Atorvastatin on the Pharmacokinetics of Co-administered Drugs - atorvastatin tablet 04
This is a table outlining the effects of different co-administered drugs and dosing regimens on the drug Atorvastatin. The table lists the name and dose of each drug, as well as percentage changes in AUC and Gy for Atorvastatin after the co-administration. The drugs included in the table are Avtipyrine, Digoxin, Oral contraceptive with norethindrone and ethinlestradiol, Tipranavir with ritonavir, Fosamprenavir with ritonavir.*
Figure 1: Effect of Atorvastatin Calcium 10 mg/day on Cumulative Incidence of Non Fatal Myocardial Infarction or Coronary Heart Dis ease Death (in ASCOT-LLA) - atorvastatin tablet 05
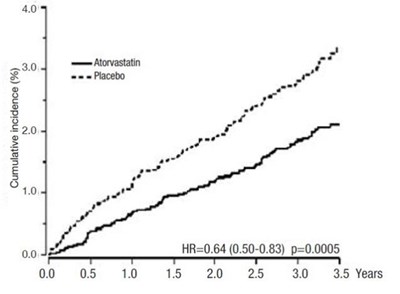
The data provided is a table or graph showing the cumulative incidence in percentage over a period of 35 years. The graph includes data related to the use of Atorvastatin, where a HR (Hazard Ratio) of 0.64 was observed. However, due to lack of context, it is not possible to explain the significance of the data.*
Figure 2: Effect of Atorvastatin Calcium 10 mg/day on Time to Occurrence of Major Cardiovascular Event (myocardialinfarction, acute CHD death, unstable angina, coronary revascularization, or stroke) in CARDS - atorvastatin tablet 06
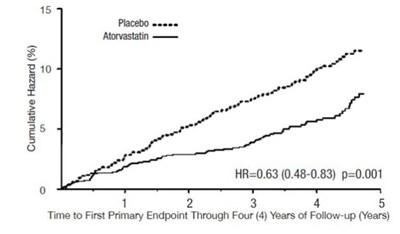
This text displays a cumulative hazard chart with time to the first primary endpoint through four years of follow-up. The hazard ratio (HR) with 95% confidence interval (0.63 (0.48-0.83)) and p-value (p=0.001) are also provided.*
Figure 3: Effect of Atorvastatin Calcium 80 mg/day vs. 10 mg/day on Time to Occurrence of Major Cardiovascular Events (TNT) - atorvastatin tablet 07
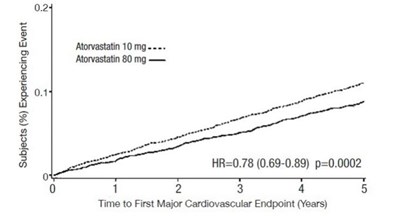
The text presents a chart showing the percentage of subjects who experienced an event during a study with two different dosages of Atorvastatin (10mg and 80mg) and the corresponding results in hazard ratio and p-value. It also includes a time axis with five intervals (years) for the occurrence of a major cardiovascular endpoint.*
TABLE 6. Overview of Efficacy Results in TNT - atorvastatin tablet 08
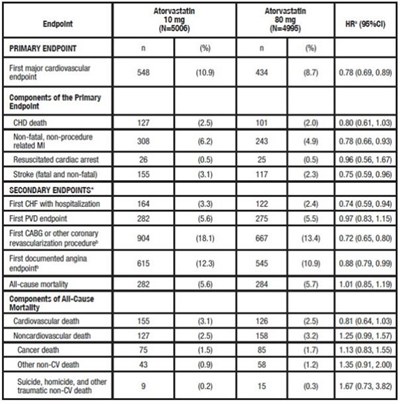
This appears to be a medical study report detailing the primary and secondary endpoints of the study. It includes information about the specific components of the primary endpoint, which are CHD death, non-fatal non-procedure related M, resuscitated cardiac arrest, and stroke. It also includes information on the first CHF with hospitalization, first PVD endpoint, first documented angina endpoint, cardiovascular death, non-cardiovascular death, and suicide, homicide, and other traumatic non-CV deaths as secondary endpoints. The numbers in parentheses appear to be statistical values or confidence intervals.*
TABLE 7. Dose Response in Patients With Primary Hyperlipidemia (Adjusted Mean % Change From Baseline)a - atorvastatin tablet 09
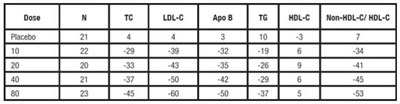
TABLE 8. Mean Percentage Change From Baseline at Endpoint (Double-Blind, Randomized, Active-Controlled Trials) - atorvastatin tablet 0a
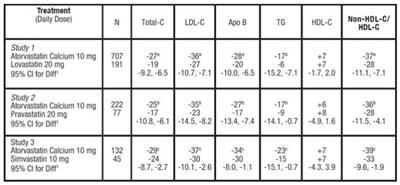
This text appears to be reporting the results of a study comparing the effects of various doses of different medications to treat a condition. The medications mentioned are Atorvastatin, Lovastatin, Rosuvastatin, Pravastatin, and Simvastatin. The study reports the dosage of each medication used, the number of participants, and the 95% confidence interval for the difference in efficacy between the medications.*
TABLE 9. Combined Patients with Isolated Elevated TG: Median (min, max) Percentage Change From Baseline - atorvastatin tablet 0b
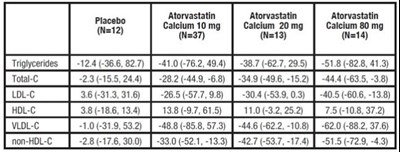
This appears to be a table of numerical data related to atorvastatin, a medication used to lower cholesterol and triglyceride levels. The table includes values for calcium intake and the number of participants (N) for each level of calcium intake. There are also measurements for triglyceride levels, HDL-C levels, and non-HDL-C levels, as well as confidence intervals for each value. Without further context, it is difficult to draw any definitive conclusions from these numbers.*
TABLE 10. Open-Label Cross over Study of 16 Patients With Dysbetalipoproteinemia (Fredricks on Type III) - atorvastatin tablet 0c
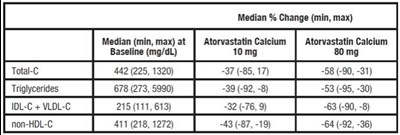
This appears to be data related to the medication Atorvastatin Calcium. It shows the median percentage change in different measurements such as Median (min, max) at Baseline (mg/dL) for different dosages of the medication. The text also contains information about different measurements such as Triglycerides and non-HDL-C.*
TABLE 11. Lipid-altering Effects of Atorvastatin Calcium in Adolescent Boys and Girls with Heterozygous Familial Hypercholesterolemia or Severe Hypercholesterolemia (Mean Percentage Change From Baseline at Endpoint in Intention-to-Treat Population) - atorvastatin tablet 0d
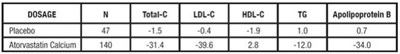
This text appears to be tabular data showing dosage information for two different treatments. The first treatment is a placebo, while the second treatment is Atorvastatin Calcium. The data includes measurements for various cholesterol components (Total-C, LoL-C, and HOL-C) as well as T6 and Apolipoprotein B. Without further context, the purpose of this data is not clear.*
PRINCIPAL DISPLAY PANELNDC 10135-0649-10Atorvastatin Calcium Tablets10 mg1000 TABLETSRx Only - atorvastatin tablet 0e

PRINCIPAL DISPLAY PANELNDC 10135-0650-05Atorvastatin Calcium Tablets20 mg500 TABLETSRx Only - atorvastatin tablet 0f

This is a medication called DMARLEX Atorvastatin, manufactured by Marlex Pharmaceuticals, Inc. Each film-coated tablet contains 20 mg Atorvastatin calcium. The usual dosage and prescribing information should be followed. The medication should be stored in a tight, child-resistant container at a temperature between 20° to 25°C. The accompanying patient information should be dispensed by the pharmacist to each patient.*
PRINCIPAL DISPLAY PANELNDC 10135-0651-10Atorvastatin Calcium Tablets40 mg1000 TABLETSRx Only - atorvastatin tablet 10

This is a medication with NDC code 10135-0651-10. Each tablet contains 40mg of atorvastatin calcium. The medication is usually prescribed and should be stored at 20°-25°C. The tablets should be dispensed in a child-resistant container and kept out of the reach of children. The accompanying patient information should be dispensed to each patient by the pharmacist. This medication is manufactured and distributed by Marlex Pharmaceuticals, Inc. with headquarters in New Castle, DE.*
atorvastatin tablet 11

This is a medication with the NDC code 10135-0653-05. Each film-coated tablet contains 80mg of atorvastatin calcium equivalent to atorvastatin. The recommended dosage is indicated in the accompanying prescribing information. The medication should be stored between 20-25°C (68-77°F). The medication should be dispensed in a tight, child-resistant container and kept out of reach of children. Patient information should also be provided to each patient. The medication is manufactured and distributed by Marlex Pharmaceuticals, Inc. This package contains 500 tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.