Product Images Alendronate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Alendronate NDC 10135-696 by Marlex Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The molecular formula of alendronate sodium is C4H12NNaO7P2•3H2O and its formula weight is325.12. The structural formula is: - alendronate sodium tablets 01

Not available.*
Figure 1:Cumulative Incidence of Hip Fractures in the Three-Year Study of FIT (patients with radiographic vertebral fracture at baseline) - alendronate sodium tablets 02

Figure 2: Osteoporosis Treatment Studies in Postmenopausal WomenIncrease in BMD Alendronate 10 mg/day at Three Years - alendronate sodium tablets 03
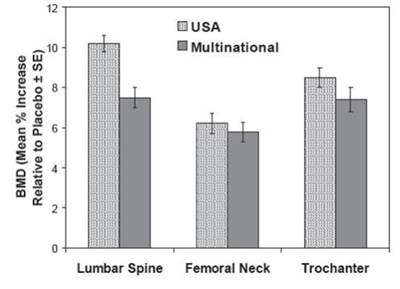
Figure 3: Figure 3: Osteoporosis Treatment Studies in Postmenopausal WomenTime Course of Effect of Alendronate 10 mg/day Versus Placebo: Lumbar spine BMD Percent Change From Baseline - alendronate sodium tablets 04

Figure 4 - alendronate sodium tablets 05

The text contains a table with the columns "Change in BMD from Baseline 2-Year Study" and "Change in BMD from Baseline 3-Year Study" and rows of data labelled "Alendronate Sodium 5mg/day" and "Placebo". The table shows percentages of mean change in BMD in different parts of the body including spine, neck, body, femoral trochanter, and total. There is no clear description or purpose provided for this data.*
Figure 5 - alendronate sodium tablets 06

The text appears to be a table or chart showing the increase in bone mineral density (BMD) for patients with glucocorticoid treatment who were given the medication Alendronate at a dose of 5 mg/day for one year. The table shows the mean 9% increase in BMD relative to a placebo, with separate increases shown for the lumbar spine, femoral neck, and trochanter. This information is likely from a clinical trial or study evaluating the efficacy of Alendronate in improving BMD in patients with glucocorticoid treatment.*
Figure 6: - alendronate sodium tablets 07

This text appears to be a part of a study on Paget's disease of bone. The study evaluates the effect of Alendronate 40mg/day on serum alkaline phosphatase compared to Placebo or Etidronate 400mg/day. The study is a multinational trial and a US Study. The graph shows the results at different time intervals (in months).*
PRINCIPAL DISPLAY PANEL- 35 mg Tablet Blister Pack Carton4 TabletsRx onlyNDC 10135-0696-44Once Weekly - alendronate sodium tablets 08

PRINCIPAL DISPLAY PANEL- 35 mg Tablet Blister Pack Carton12 (3×4) Unit-of-Us e TabletsRx onlyNDC 10135-0696-12Once Weekly - alendronate sodium tablets 09

This is a description for Alendronate Sodium Tablets, USP 35 mg. It is a weekly medication for adults who suffer from osteoporosis. It contains 12 tablets per package and each tablet is 35mg in strength. The recommended dosage is one 35mg tablet per week, accompanied by a glass of water. The drug comes with a prescription label and should only be dispensed by a pharmacist. The medication is manufactured by Mtx Pharmaceuticals in New Castle, CA and distributed in the USA.*
PRINCIPAL DISPLAY PANEL- 70 mg Tablet Blister Pack Carton4 TabletsRx onlyNDC 10135-0697-44Once Weekly - alendronate sodium tablets 0a

This appears to be a product label for Alendronate Sodium Tablets, used to treat bone loss. The label includes dosage instructions, manufacturer information, and warnings about potential side effects. It is recommended to store the medication at room temperature.*
PRINCIPAL DISPLAY PANEL- 70 mg Tablet Blister Pack Carton12 (3×4) Unit-of-Use TabletsRx onlyNDC 10135-0697-12 Once Weekly - alendronate sodium tablets 0b

This appears to be a description of a medication called "Alendronate Sodium Tablets". Each tablet contains 70mg of the active ingredient, sodium alendronate, and is meant to be taken once a week as indicated on the prescription label. The manufacturer is listed as Marx Pharmaceuticals and the tablets come in a pack of 12. The rest of the text is illegible or fragmented and is not-usable to generate any further information.*
PRINCIPAL DISPLAY PANEL- 70 mg Tablet Blister Pack Carton20 (2×10) Unit-Dose TabletsRx onlyNDC 10135-0697-20Once Weekly - alendronate sodium tablets 0c

This is the description: "Once Weekly Alendronate Sodium Tablets, USP 70 mg is a medication manufactured by Marx Pharmaceuticals. It comes in tablet form and is intended for adults. The usual adult dosage of this medication is one 70 mg tablet once a week. It contains 1.37 mg of alendronate sodium per tablet. This medication requires a prescription label. For further information, refer to the enclosed medication guide. Keep all drugs out of reach of children and store at 20°-25°C (68-77°F) with excursions permitted to 15°-30°C (59°-86°F)."*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.