Product Images Desvenlafaxine
View Photos of Packaging, Labels & Appearance
- 821-10 - 821 10
- 0821-14 - 821 14
- 821-30 - 821 30
- 821-90 - 821 90
- 822-10 - 822 10
- 822-14 - 822 14
- 822-30 - 822 30
- 822-90 - 822 90
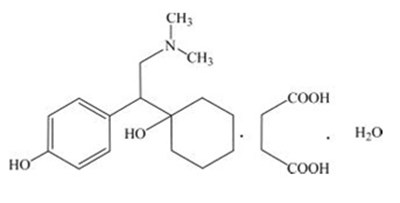
- DEVStructure - DEVStructure
- Fig4-Study5Relapsevs.randomization - Figure4
- Fig5-Study6-Relapsevs.Randomization - Figure5
- figure2 - fig2final2
- figure1 - figure1
- figure3 - figure3
Product Label Images
The following 14 images provide visual information about the product associated with Desvenlafaxine NDC 10135-822 by Marlex Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
821-10 - 821 10

This text provides information about Desvenlafaxine Extended-Release Tablets, each containing 76mg desvenlafaxine succinate equivalent to 50mg desvenlafaxine. It gives directions for use, stating to swallow whole and not cut, crush, or chew the tablets. The storage conditions are also provided, recommending storing between 20°C to 25°C with excursions permitted between 15°C to 30°C. The tablets are to be kept out of the reach and sight of children. The manufacturer is Marlox Pharmaceuticals, Inc. located in New Castle, DE. The package contains 1000 tablets of 50mg each.*
821-30 - 821 30

This is a description of medication with NDC 10136-0821. Each extended-release tablet contains 76 mg of desvenlafaine succinate equivalent to 50 mg of desvenlafaxine. It is recommended to store the tablets at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F). The medication should be kept out of reach and sight of children. The packaging contains 30 tablets.*
821-90 - 821 90

Each extended-release tablet contains 76 mg desvenlafaine succinate equivalent to 50 mg Geovaaimine B. The usual dosage instructions can be found on the package insert. It is important not to swallow whole, crush, dissolve, or chew the extended-release tablet. Store between 15° to 30°C (59° to 86°F). Keep out of reach and sight of children. This medication is prescription only.*
822-10 - 822 10

Each extended-release tablet contains 152 mg desvenlafaxine succinate equivalent to 100 mg desvenlafaxine. It is recommended to swallow the tablet whole without splitting, crushing, or chewing. The tablets should be stored at 20°C to 25°C (68°F to 77°F) with excursions between 15°C to 30°C (59°F to 85°F) permitted. Keep the medication out of reach of children. This product is manufactured for distribution by Marlex Pharmaceuticals, Inc. in New Castle, DE. The label indicates it is Desvenlafaxine Extended-Release Tablets. It is advised for pharmacists to dispense the medication using the unit dose provided separately for each patient. This container holds 1000 tablets.*
822-90 - 822 90

Each extended-release tablet contains 152 mg of desvenlafaxine succinate, equivalent to 100 mg of desvenlafaxine. These tablets should be swallowed whole and not split, crushed, or chewed. The tablets should be stored at a temperature between 20°C to 25°C, with excursions permitted between 15°C to 30°C. Keep out of reach of children. This product is manufactured for/distributed by Marlex Pharmaceuticals. Pharmacists should dispense the medication as prescribed. This is a prescription-only medication.*
Fig4-Study5Relapsevs.randomization - Figure4

This text provides information on the estimated proportion of patients experiencing a relapse over time, showing values at different intervals such as 80%, 40%, 30%, 20%, and 10%. The table also includes a timeline for when relapses occur, measured in days from randomization, comparing Desvenlafaxine Extended-Release Tablets 50mg to Placebo.*
Fig5-Study6-Relapsevs.Randomization - Figure5

This text provides information on the estimated proportion of patients experiencing relapse over time, as well as the time to relapse in days from randomization. The data seems to compare a medication called Desvenlafaxine Extended Release Tablets with a placebo, but additional details and context are needed for a complete evaluation.*
figure2 - fig2final2
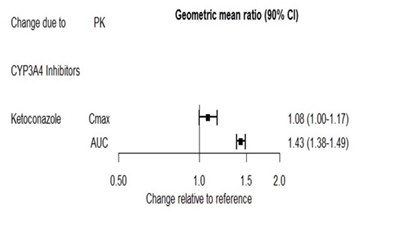
This text provides information about the effect of CYP3A4 inhibitors, specifically ketoconazole, on geometric mean ratios for Cmax and AUC. The values for Cmax and AUC are shown with their corresponding 90% confidence intervals. These values are compared against a reference for relative change.*
figure1 - figure1

This is an evaluation of the geometric mean ratio with 90% Confidence Intervals for different levels of renal impairment and hepatic impairment. The data includes comparisons for mild, moderate, and severe cases, as well as for End Stage Renal Disease (ESRD) and hepatic impairment. The results are presented for parameters like Cmax (maximum concentration) and AUC (area under the curve). Additionally, information is provided for specific populations, like females and different age groups. The analysis includes changes relative to a reference point and clarifies the abbreviation "ESRD" as End Stage Renal Disease.*
figure3 - figure3

This passage appears to be a table showing the effects of interacting drugs on certain substrates and metabolic pathways. It includes data such as changes in concentration levels (Cmax), area under the curve (AUC), and other pharmacokinetic parameters. The text discusses drug interactions involving Desvenlafaxine, Desipramine, Midazolam, Tamoxifen, and Aripiprazole, among others. The information provided may be useful for understanding how these drugs interact and affect each other's metabolism in the body.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.