FDA Label for Blistex European Formula
View Indications, Usage & Precautions
Blistex European Formula Product Label
The following document was submitted to the FDA by the labeler of this product Blistex Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
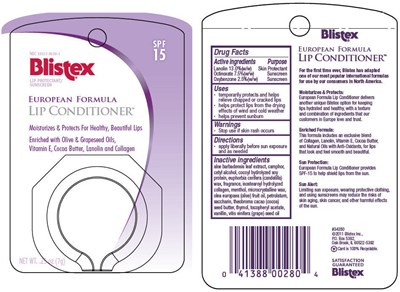
Drug Facts
#34280
©2011 Blistex Inc.,
P.O. Box 5392,
Oak Brook, IL 60522-5392
Otc - Purpose
| Active ingredients | Purpose |
|---|---|
| Lanolin 13 .0%(w/w) | Skin Protectant |
| Octinoxate 7.5%(w/w) | Sunscreen |
| Oxybenzone 2.5%(w/w) | Sunscreen |
Uses
- temporarily protects and helps relieve chapped or cracked lips
- helps protect lips from the drying effects of wind and cold weather
- helps prevent sunburn
Warnings
- Stop use if skin rash occurs
Directions
- apply liberally before sun exposure and as needed
Inactive Ingredients
aloe barbadensis leaf extract, camphor, cetyl alcohol, cocoyl hydrolyzed soy protein, euphorbia cerifera (candelilla) wax, fragrance, isostearoyl hydrolyzed collagen, menthol, microcrystalline wax, olea europaea (olive) fruit oil, petrolatum, saccharin, theobroma cacao (cocoa) seed butter, thymol, tocopheryl acetate, vanillin, vitis vinifera (grape) seed oil
Principal Display Panel - 7 G Container Label
NDC 10157-9539-1
Blistex®
LIP PROTECTANT/
SUNSCREEN
SPF
15
EUROPEAN FORMULA
LIP CONDITIONER™
Moisturizes & Protects For Healthy, Beautiful Lips
Enriched with Olive & Grapeseed Oils,
Vitamin E, Cocoa Butter, Lanolin and Collagen
NET WT. .25 oz (7g)
* Please review the disclaimer below.