FDA Label for Carmex Cold Sore Treatment External Analgesic Skin Protectant
View Indications, Usage & Precautions
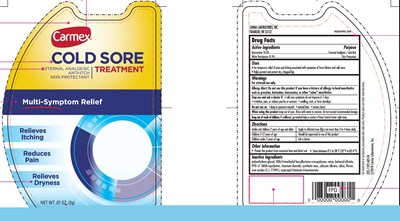
Carmex Cold Sore Treatment External Analgesic Skin Protectant Product Label
The following document was submitted to the FDA by the labeler of this product Carma Laboratories, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredients
Benzocaine 10.0%
White Petrolatum 45.9%
Purpose
External Analgesic / Anti-Itch
Skin Protectant
Uses
- For temporary relief of pain and itching associated with symptoms of fever blisters and cold sores
- Helps prevent and protect dry, chapped lips
Warnings
For external use only.
Allergy Alert: Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics
Stop Use And Ask A Doctor If
- cold sore symptoms do not improve in 7 days
- irritation, pain, or redness persists or worsens
- swelling, rash, or fever develops
Do Not Use On
• deep or puncture wounds • animal bites • serious burns
When Using This Product
keep out of eyes. Rinse with water to remove. Do not exceed recommended dosage.
Keep Out Of Reach Of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 2 years of age and older | Apply to affected area (lips) not more than 3 to 4 times daily |
| Children 2-12 years of age | Should be supervised in use of this product |
| Children under 2 years of age | Ask a doctor |
Other Information
- Protect this product from excessive heat and direct sun
- Store between 0°C to 28°C (32°F to 82.4°F)
Inactive Ingredients
polyethylene glycol, HDI/trimethylol hexyllactone crosspolymer, mica, behenyl olivate, PPG-51 SMDI copolymer, titanium dioxide, synthetic wax, calcium silicate, silica, flavor, iron oxides (C.I. 77491), isopropyl titanium triisostearate
Package Labeling:
* Please review the disclaimer below.