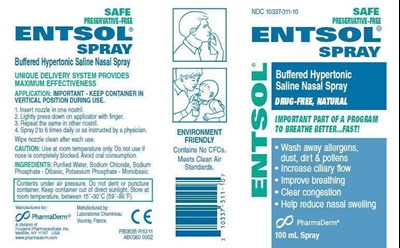
FDA Label for Entsol
View Indications, Usage & Precautions
Entsol Product Label
The following document was submitted to the FDA by the labeler of this product Pharmaderm A Division Of Fougera Pharmaceuticals Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient:
Sodium chloride (3%)
Purpose:
Moisturizer
Inactive Ingredients
Purified Water, Sodium Phosphate - Dibasic, Potassium Phosphate - Monobasic.
Package Label – Principal Display Panel – Container
NDC 10337-311-10
Package Label – Principal Display Panel – Carton
NDC 10337-311-10
* Please review the disclaimer below.