Product Images Oxycodone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Oxycodone Hydrochloride NDC 10702-056 by Kvk-tech,inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 1 chemical structure
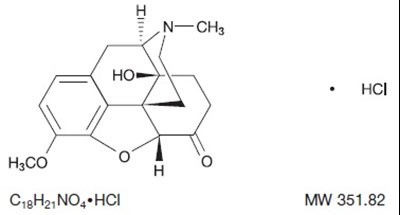
This appears to be a chemical compound described by its molecular formula, MW (molecular weight) and an acronym. The acronym "Hol" and "HsCO o o" are not clear and may be due to errors. The compound is "C1sHaNO,HCI" with a molecular weight of 351.82. Additional information may be required to determine the specific characteristics and properties of this compound.*
figure 10 20mg 100s

This is a medication information sheet for Oxycodone Hydrochloride Tablets, USP containing 100 tablets. The medication is for prescription use only and the recommended dosage should be followed as per the package or as recommended by the healthcare professional. The text also mentions storage, tamper evident packaging and advises the pharmacist to dispense an accompanying medication guide to each patient.*
figure 11 20mg 500s

This is a description for a medication called "Oxycodone Hydrochloride Tablets, USP". It comes in a bottle containing 500 tablets. The recommended dosage is specified on the package. The medication should be stored within a temperature range of 15-30°C and kept out of reach from children. The accompanying Medication Guide should be dispensed to each patient. The rest of the text seems to be identifying codes and not relevant information.*
figure 13 30mg 500s

This is a pharmaceutical label displaying two National Drug Codes. One of them is 10702-009-50 and the other is 1070200850. The tablet contains Oxycodone, Dipane Hydrochloride and other ingredients. The recommended dosage information is available on the packaging. The label also includes some instructions for pharmacists and some prescription details. The text towards the end is unclear and not legible.*
figure 2 fig1 graph

The text describes a dose-proportionality study of Oxycodone Hydrochloride in three different doses: 5mg, 15 mg, and 30 mg. The study involved a single-dose of these three levels, with multiple observations at each level. The results of the study are presented graphically in a figure showing the concentration (in ng/mL) of Oxycodone Hydrochloride over time (in hours).*
figure 4 5mg 100s
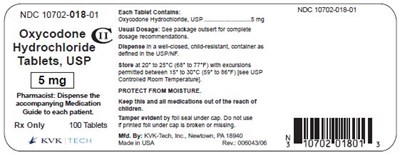
This is a label for Oxycodone Hydrochloride Tablets, USP with 100 tablets per package. The package includes a medication guide for use by the patient. The usual dosage is indicated on the package. The tablets should be kept at room temperature and protected from tampering.*
figure 5 5mg 500s

The text seems to be a medication label or prescription for Oxycodone Hydrochloride Tablets, USP with the dosage of "500 Tablets". It also includes a reference code "NDG 1070201850" and "710702 01850". The label contains instructions for the pharmacist to provide medication guidance to the patient.*
figure 6 10mg 100s
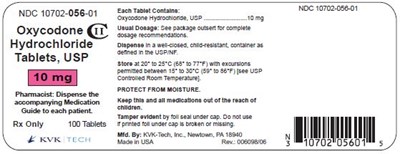
This is a medication called Oxycodone Hydrochloride in tablet form that is sold only with a prescription to treat moderate to severe pain. The pharmacist must provide a medication guide to each patient. It comes in a pack of 100 tablets and should be stored at room temperature, out of reach of children. The recommended dose can be found on the package or as directed by a doctor. The medication has a specific code number (NDC) for identification purposes.*
figure 7 10mg 500s

This is a medication package containing Oxycodone hydrochloride tablets. The tablets are in a bottle with NDC number 1070206680 and are used to relieve moderate to severe pain. The medication must be protected from moisture and is dispensed through a prescription. A guide to the medication has been provided to each patient. The manufacturer is located in Newtown, A15640. No further information is available as the text contains several errors.*
figure 8 15mg 100s

This is a description for medication named "Oxycodone Hydrochloride Tablets, USP". The medication is available in 100 tablets and requires a prescription to dispense. The pharmacist must give a medication guide to each patient. The package contains complete dosage information. The medication must be kept in a cool and dry place between 12-25 °C and kept away from light. The medication comes in a tamper-proof container with an under cap seal.*
figure 9 15mg 500s

The text appears to be a description of a medication called Oxycodone Hydrochloride Tablets, USP with NDC number 10702-008-50. This medication is Rx Only and comes in a package of 500 tablets. Each tablet contains Syt o and has a recommended dosage of 15y. The pharmacist is instructed to dispense the medication guide that accompanies each package to the patient. The text includes other details such as instructions on storage and tamper evidence, but some parts are not readable due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

