Product Images Oxycodone Hydrochloride
View Photos of Packaging, Labels & Appearance
- chemical structure - figure 01 chemical structure
- logo - figure 02 kvk logo
- syringe - figure 03 syringe
- 5 mg container label - figure 04 5 mg container label
- 100 mg container label - figure 05 100 mg container label
- 5mg/5mL Carton - figure 06 5 mg carton label
- 100mg/5mL Carton - figure 07 100 mg carton label
Product Label Images
The following 7 images provide visual information about the product associated with Oxycodone Hydrochloride NDC 10702-183 by Kvk-tech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
syringe - figure 03 syringe

This is a set of instructions on how to measure the dose from a syringe plunger. The instructions advise measuring the dose from the widest portion of the plunger and not the narrow tip. There are several symbols and characters that cannot be interpreted, but they do not affect the overall meaning of the text.*
5 mg container label - figure 04 5 mg container label

This text is a mixture of information about a medication called Oxycodone Hydrochloride Oral Solution. It includes details about its NDC code (10702-183-50), concentration (1 mg per 1 mL), dosage, administration, storage, and packaging. Additionally, it mentions a medication guide that needs to be provided to each patient, and some warnings about keeping it away from children. However, there are some sentences that are not readable, and the has produced some errors in the text, making it difficult to provide a complete and reliable description.*
100 mg container label - figure 05 100 mg container label

This is a medication label for Oxycodone Hydrochloride in Oral Solution form, with a prescribed dosage of 100 mg per 5 ml or 20 mg per 1 ml. The label instructs the pharmacist to dispense the medication with an accompanying Medication Guide to the patient. The NDC number is 1070247103 and the Rx is only meant for the person it is prescribed to. The text also shows a NOC number of 0217108.*
5mg/5mL Carton - figure 06 5 mg carton label

This is a description of a medication called Oxycodone Hydrochloride Oral Solution. It is available as a 5mg per 5(1 mg per 1 mL) oral solution in a 500mL bottle. The inactive ingredients are glycerin, malitol syrup, sodium benzoate, sucralose powder, hydrochloric acid, sodium hydroxide, sorbiol, natural and artificial berry flavor, FD&C Red #40 powder, and purified water. The medication guide is enclosed in the packaging and contains prescribing information. The medication should be dispensed in the provided bottle with the calibrated measuring cup. It is advised to store the medication between 15°C to 30°C (59°F to 86°F) and protected from moisture, light, and sugar. The medication does not contain any ingredient made from gluten-containing grains.*
100mg/5mL Carton - figure 07 100 mg carton label
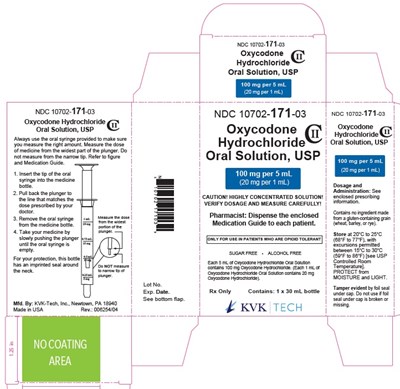
This is a medication label for Oxycodone Hydrochloride Oral Solution, USP including dosage and administration guidance. It also includes instructions for using the provided oral syringe to measure the right amount and cautions against using any other method. The label also includes information about the manufacturer and lot number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

