Product Images Up And Up Miconazole 1
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Up And Up Miconazole 1 NDC 11673-737 by Target Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Up & Up Miconazole 1 Image 2 - 01 30 17 73745UWC1 Image 1

This is a medicine for the treatment of vaginal yeast infections. It contains two active ingredients, one for vaginal ingestion and one in external cream form. It warns against using it if one is experiencing symptoms for the first time, if there are other symptoms present such as lower abdominal pain, if exposed to HIV or if also taking the prescription blood-thinning medicine warfarin, among others. It provides directions for use and instructs not to use for children under 2 years of age. However, manufacturer information is unreadable.*
Up & Up Miconazole 1 Image 1 - 01 30 17 73745UWC1 Image 2

This is a product description for Up&Up, a vaginal antifungal treatment that compares to the active ingredient in Monistat 1. The combination pack comes with one vaginal softgel insert and one tube of external cream, and three feminine wipes with aloe and vitamin E. The product helps cure most vaginal yeast infections and relieves associated external itching and irritation. The NDC number is 11678-737-45.*
Applicator.jpg - image 02
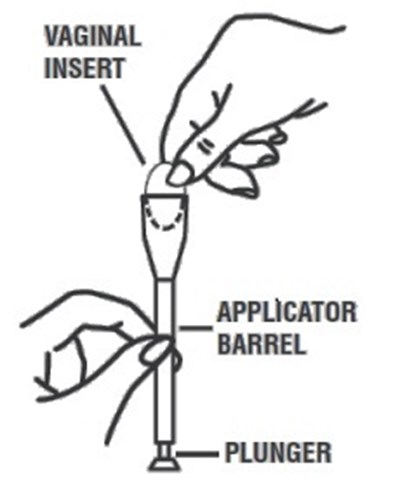
This text is describing a vaginal insert that comes with an applicator barrel and a plunger.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


