Product Images Sublocade
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 20 images provide visual information about the product associated with Sublocade NDC 12496-0100 by Indivior Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
sub0a 0038 14

The text describes a chart showing the percentage of subjects at different levels (100%, 80%, 60%, 40%, 20% and 0%) in a clinical trial for a medication called SUBLOCADE, in two different doses and with an IDC component. The first dose is 300mg/300mg, the second is 300mg/100mg, and the control is a placebo with IDC. The chart seems to be measuring something related to the number of opioid-free weeks.*
sub0a 0038 15

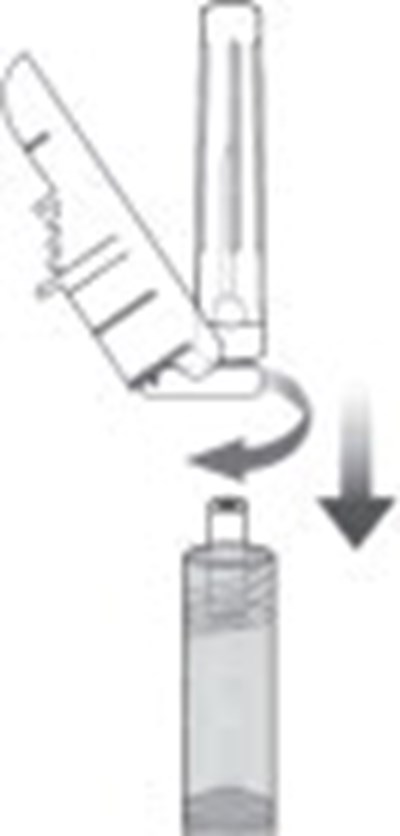
This is a prescription for Sublocade, a subcutaneous injection used for abdominal pain. The users are warned to read all the instructions carefully before the usage of the injection. The needle should be attached during administration, and the needle provided with the injection should only be used. The injection must be kept out of reach of children and should be stored at a temperature between 29-8°C (35.6°-46.4°F). The users must remove the Sublocade from the refrigerator 15 minutes before administering it. There is a warning that serious harm or death can occur if it is injected intravenously. It is also mentioned that after 7 days, the injection should be discarded if kept outside the refrigerator. For more information, SUBLOCADE com can be visited.*
sub0a 0038 16

Sublocade' @ is a single dose injection used for abdominal subcutaneous injection only. It contains 100mg/0.5mL of Buprenorphine in the ATRIGEL Delivery System and an oxygen absorber. It is manufactured by Indivior Inc. and distributed by AMRI, Burlington. The product needs to be stored at 2°-8°C and discarded if left at room temperature for more than 7 days. The needle should be attached at the time of administration, and only the provided needle should be used. The expiry date for LOTX00 is 12/.*
sub0a 0038 17

Sublocade is a sterile single-dose injection used for subcutaneous use. It contains buprenorphine extended-release and is only available with a prescription. The product has a barcode and a manufacturing location in Massachusetts. The lot number and expiration date are also provided, but there is no available information regarding the dosage or quantity as they are not mentioned in the provided text.*
sub0a 0038 18

Sublocade is a medication that comes in injection form and is used only for abdominal subcutaneous injection. It should be kept out of reach of children. The accompanying full prescribing information needs to be read prior to use. Serious harm or death could result if injected intravenously. The product needs to be removed from the fridge at least 15 minutes before administration and must reach room temperature to be used. The needle provided should be attached at the time of administration and stored between 35.6°F and 46.4°F. If left at room temperature for over 7 days, it should be discarded.*
sub0a 0038 19

Sublocade @ is a sterile single-dose injection used for abdominal or subcutaneous administration with 300 mg/1.5 mL of buprenorphine in the ATRIGEL® delivery system per prefilled syringe. The product should be stored between 2°-8°C (35.6°-46.4°F), but may be kept at room temperature for up to 7 days prior to administration. After this time, the product should be discarded. The needle provided should be used for administration after the product has been removed from the refrigerator and allowed to warm to room temperature for at least 15 minutes. This extended-release medication is to be used only as prescribed and kept away from children.*
sub0a 0038 20

This is a description of a prescription medication with the NDC code 12496-0300-5 called Sublocade, which is a buprenorphine extended-release injection for subcutaneous use. The medication is for subcutaneous injection only and is designed for single dose use. There is a lot number (XXXXXXXX) and an expiration date (1272017) provided. The manufacturer is listed as AMRI with an address in Burlington, MA. A linear barcode is present as well as an unvarnished area for wrap.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.