Product Images Hydrocodone Bitartrate And Acetaminophen
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Hydrocodone Bitartrate And Acetaminophen NDC 12634-978 by Apotheca, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 325mg Amneal - 10 325mg Amneal

This text is a description of a prescription medication named Hydrocodone Bitartrate and Acetaminophen 10/325mg. It is repackaged and distributed by Apotleca in Arizona, and is available in a container that is child-resistant as per the guidelines of USP. The text includes details on NDC, lot number, and expiration date. It also includes directions for use, storage, and precautions that must be taken while handling.*
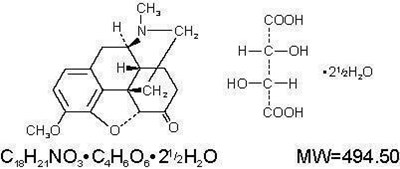
image of formula - hydro 02

The text provided represents two chemical formulas: CH4NOH and CHNO. The molecular weight of the latter is 151.16. It is unclear from the text what the context or purpose of these formulas may be.*
hydro 04 - hydro 04

This is a medication description for tablets. The tablets contain hydrocodone bitartrate and acetaminophen in the strength of 10mg and 500mg, respectively. The usual adult dosage is one tablet every four to six hours, as needed, for pain. The total daily acetaminophen dosage should not exceed six tablets. The tablets should be dispensed in a tight, light-resistant container with a child-resistant closure. The tablets are manufactured by Watson Laboratories, Inc. and repackaged and distributed by Apotheca, Inc. The expiration date and lot number are not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.