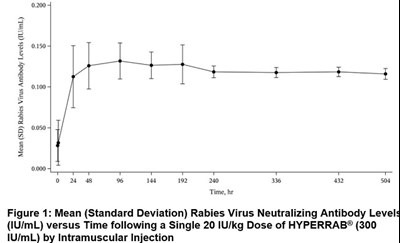
Product Images Hyperrab
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Hyperrab NDC 13533-318 by Grifols Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1 ml carton - 1mLcarton

Rabies immune globulin is a sterile solution of immunoglobulin containing 15%-18% protein stabilized with .16 10 0.26 M glycine. Each vial contains 300 1U/mL of potency based on the U.S. standard for rabies immune globulin. This product is for infiltration and intramuscular injection only and is not to be given intravenously. The patient and physician are advised to discuss the risks and benefits of this product. This solution is suitable for use with rabies vaccine. It must be stored at 2-8°C (@6-46°F) and must be used or discarded after the expiration date. The product is not returnable for credit or exchange.*
1 ml vial - 1mlvial

This is a description of a product. The product is a single-dose vial of Rabies Immune Globulin (Human) and has the National Drug Code (NDC) of 13533-318-10. The text includes a warning to not give the product intravenously and to discuss the risks and benefits with a healthcare provider. The product should be discarded if there is any unused portion. The use directions are also provided. The manufacturer's address is listed as TP, NC 27700 USA.*
3 ml carton - 3mLcarton

This text contains information about Rabies Immune Globulin, a sterile solution used for infiltration and intramuscular administration. It contains about 169-18% protein stabilized, and should be kept below 25°C (77°F) for six months after any use. There are instructions for dosage and administration, which should be read before use, and it is only suitable for use with Rabies Vaccine. The potency of the solution is 300 IU/mL and it is made by Grifols Therapeutics LLC, located in North Carolina, USA.*
3 m vial - 3mlvial

This is a description of a single-dose vial of Rabies Immune Globulin (Human). The dosage recommended is 20 IU/kg given intramuscularly (into the skin) for the treatment of rabies infection. The product should not be given intravenously. The remaining product should be discarded after use. This product is licensed under number 1871 B.*
5 ml carton - 5mLcarton

This is a description of a medicine called Rabies Immune Globulin. It is a single-dose vial containing a sterile solution of immunoglobulin with a potency of 300 IU/mL. The solution is stabilized with glycine and is not made with natural rubber. The medication is only for intramuscular injection and not to be given intravenously. The packaging contains more information on administration, and proper storage and disposal. The manufacturer is Grifols Therapeutics LG.*
Figure 2 - figure2
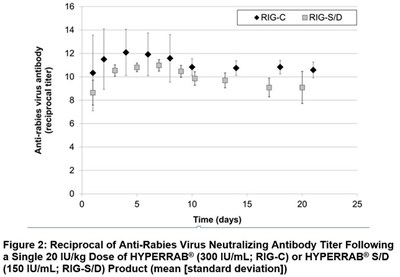
This text describes a figure labeled "Figure 2" that shows the reciprocal of the anti-rabies virus neutralizing antibody titer over time after a single dose of HYPERRAB® or HYPERRAB® S/D product. The figure also includes a time axis in days and a titer axis from 0 to 25.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.