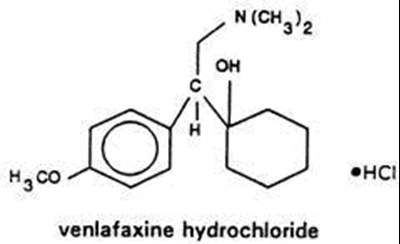
Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 13668-019 by Torrent Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - a7f843b6 0935 4be6 a465 97179a846148 01

This text includes a list of interacting drugs that may require caution when used together. The drugs mentioned are emanal, Dlazspam, Ketosonszole, Ketosonszals, wotopror, imipramine, and Litnam. The recommendation for drugs used concomitantly with Avola is to exercise caution and to monitor blood pressure. Caution is also advised with patients having hypertension, obesity, and hepatic dysfunction. Dose adjustment may be necessary for some drugs. The last line of the text is not readable.*
Figure 3 - a7f843b6 0935 4be6 a465 97179a846148 03

This appears to be a table containing information about recommended dose adjustments for certain drugs based on the patient's medical conditions such as hepatic or renal impairment. The table specifies the percentage by which the dosage should be reduced along with the maximum concentration (Cmax) and area under the curve (AUC) values. Age and gender have also been taken into consideration for dose adjustment in some cases.*
Label - a7f843b6 0935 4be6 a465 97179a846148 07

This is a description of a medication called Venlafaxine Hydrochloride Extended-release Capsules, USP. Each capsule contains 37.5mg of Venlafaxine and should be stored at a temperature between 20°-25°C (68°-77°F), with excursions permitted between 15°-30° (58°F). The medication should be dispensed in an airtight container and comes in packages of 30 capsules. The usual dosage and prescribing information can be found in the accompanying medication guide. The medication is available only with a prescription.*
label - a7f843b6 0935 4be6 a465 97179a846148 08

This appears to be a medication label for Venlafaxine Hydrochloride, which comes in controlled-release capsules of 75 mg each and is packaged in bottles of 30. The label includes dosage instructions, storage information, and a control number. There may also be additional information provided on an accompanying prescription form.*
Label - a7f843b6 0935 4be6 a465 97179a846148 09

This text describes the medication "Venlafaxine Hydrochloride" in the form of extended-release capsules. The medication is stored at room temperature and contains FD&C Yellow No. 5 (tartrazine) as a color additive. The prescription is only available with a prescription and the pharmacist is advised to dispense the accompanying medication guide to each patient. The manufacturer of the medication is Torrent Pharmaceuticals.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.