Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
- image containerlabel 10
- image containerlabel 15
- image containerlabel 2
- image containerlabel 20
- image containerlabel 30
- image containerlabel 5
- image fig1 effectofotherdrug arip
- image fig2 effectofotherdrug dehydroarip
- image fig3 effectofarip otherdrug
- image fig4 effectofintrinsicfactor arip
- image fig5 effectofintrinsicfactor dehydroarip
- image fig6 schizophreniastudy 5
- image fig7 bipolarstudy 7
- image fig8 bipolarstudy 8
- image fig9 tourettesdisorderstudy1
- image logo medguide
- image logo pi
- image structure
Product Label Images
The following 18 images provide visual information about the product associated with Aripiprazole NDC 13668-217 by Torrent Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image containerlabel 10

Aripiprazole is a medication available in the form of tablets. Each tablet contains 10mg of USP aripiprazole. It is recommended to store it in a tight container with child-resistant closure at 20° to 25°C (68° to 77°F) with excursions between 15°C and 30°C (59°F and 86°F) considered permissible. The usual dosage guidelines are specified in the accompanying prescribing information, and pharmacists must dispense the medication guide to each patient. The product is manufactured for Torrent Pharma Inc. and is available in packs of 30 tablets. The product is labeled for prescription use only.*
image containerlabel 15

Each tablet of Aripiprazole USP contains 15 mg of the active ingredient. It is recommended to store the medication between 20 and 25°C and preserve the container with a child-resistant closure. Torrent Pharmaceuticals is the manufacturer of the medicine, which is intended for prescription use only. The usual dosage and further instructions should be consulted through the accompanying prescribing information and medication guide. The product has been assigned an NDC identifier code.*
image containerlabel 2

Each tablet contains 2mg of Aripiprazole, USP. These 30 tablets can be found under the DG 12668-216-30 label and should be stored at temperatures between 20° and 25°C (68° to 77°F). Pharacists are advised to dispense the accompanying medication guide to each patient. This product is only available by prescription, and should be kept in a light-protected container. For more information, refer to the prescribing information.*
image containerlabel 20

Aripiprazole Tablets, USP contain 20 mg of the active ingredient and should be stored between 20°C and 25°C. They can be stored between 15°C and 30°C for limited periods. These tablets should be dispensed in a tightly sealed container with a child-resistant closure. No usual dosage is provided; however, the prescribing information can be found inside. Manufactured by Torrent Pharmaceuticals in India for Torrent Pharma Inc in Levittown, PA, these tablets come in a bottle of 30 and are only available by prescription.*
image containerlabel 30

This is a description of Aripiprazole tablets, USP used as medication for treatment. Each tablet contains 30 mg of aripiprazole. The recommended storage temperature is 20° to 25°C, with excursions permitted between 15°C and 30°C. The tablets should be dispensed in a tightly closed container with a child-resistant closure. The dosage used is as prescribed by a doctor, and the accompanying prescribing information should be consulted. Pharmacists should dispense the accompanying medication guide to each patient. The tablets are produced by TORRENT PHARMACEUTICALS LTD in India, manufactured by TORRENT PHARMA INC in Levitiown, PA 19057.*
image containerlabel 5

Each tablet of this medication contains 5 mg of aripiprazole and comes in a container with 30 tablets. It should be stored between 20°C to 25°C (68°F to 77°F) with excursions allowed between 15°C and 30°C (59°F to 86°F). The medication needs to be placed in a tight container with a child-resistant closure. The prescribing information should be consulted. The pharmacist should dispense the medication guide to each patient. The medication is manufactured for Torrent Pharma Inc. and should only be taken with a prescription.*
image fig1 effectofotherdrug arip
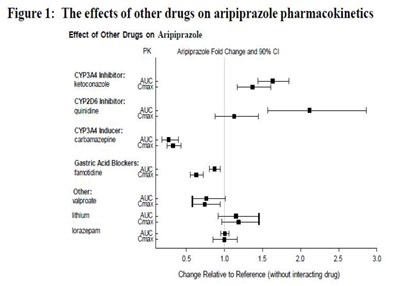
This is a figure presenting the effects of various drugs on the pharmacokinetics of aripiprazole, a medication used to treat certain mental disorders. The figure shows the fold change in aripiprazole levels and 90% confidence interval when co-administered with drugs that inhibit or induce certain metabolic enzymes. The drugs listed in the figure include ketoconazole, quinidine, carbamazepine, famidine, valproate, lithium, and lorazepam. The x-axis shows the change relative to the reference (without interacting drug) and the y-axis shows the fold change. No further information is provided.*
image fig3 effectofarip otherdrug

This is a table showing the effect of aripiprazole on the pharmacokinetics of various other drugs. The table shows fold change values and 50% confidence intervals for each drug, indicating if aripiprazole increases or decreases the drug's efficacy. There are also some drug names mentioned such as deropmetoptan, cvpace, artann, Roartain, lamotigine, valproate, lithium, lorazepam, venidaine, odesmyhenataing, and escitiopram.*
image fig4 effectofintrinsicfactor arip
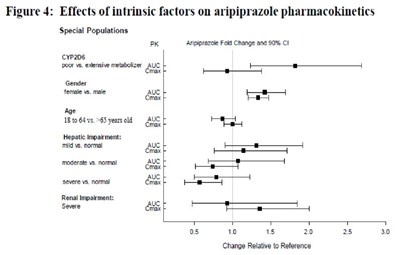
The text describes the effects of intrinsic factors on aripiprazole pharmacokinetics, including special populations such as poor vs. extensive metabolizer CYP206, female vs. male gender, and 18 to 64 years old vs. 65 years old age. It also includes the impact of hepatic impairment (mild, moderate, and severe) and renal impairment (severe). The data is presented in the form of fold change and 90% CI for AUC and Cmax.*
image fig5 effectofintrinsicfactor dehydroarip

The text describes a figure showing the effects of intrinsic factors on the pharmacokinetics of dehydro-aripiprazole, including CYP2D6 metabolizer status, gender, age, hepatic impairment severity, and renal impairment severity. The figure presents fold changes and confidence intervals for AUC and Cmax values relative to a reference group.*
image fig6 schizophreniastudy 5

This is a graph showing the proportion of patients with relapse in a schizophrenia study comparing Aripiprazole and Placebo. It includes a Kaplan-Meier estimation of cumulative proportions over time. The x-axis shows the number of days from randomization and the y-axis shows the proportion of patients with relapse. The graph also shows the number of subjects at risk for both arms.*
image fig7 bipolarstudy 7

This is a Kaplan-Meier Estimation graph showing the cumulative proportion of patients with relapse in Bipolar Study 7. The study compares Aripiprazole and Placebo. The Number of Subjects at Risk is displayed on the graph along with the number of days from Randomization.*
image fig8 bipolarstudy 8

The image shows a Kaplan-Meier estimation of the cumulative proportion of patients who experienced relapse to any mood event in Bipolar Study 8. The vertical axis shows the proportion of patients while the horizontal axis indicates the days from randomization. The drug Abilify was given to 168 patients while 169 were given placebo (LACE) out of which 77 relapsed.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




