Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
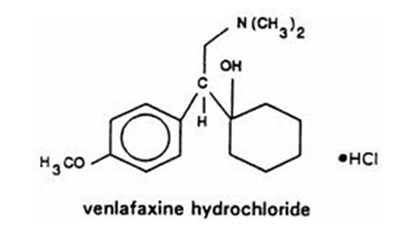
- STRUCTURAL FORMULA - venlafaxine extended release tablets 1
- 225MG 30CT - venlafaxine extended release tablets 10
- 225MG 90CT - venlafaxine extended release tablets 11
- TRIGEN LOGO - venlafaxine extended release tablets 2
- TRIGEN LOGO - venlafaxine extended release tablets 3
- 37.5MG 30CT - venlafaxine extended release tablets 4
- 37.5MG 90CT - venlafaxine extended release tablets 5
- 75MG30CT - venlafaxine extended release tablets 6
- 75MG 90CT - venlafaxine extended release tablets 7
- 150MG 30CT - venlafaxine extended release tablets 8
- 150MG 90CT - venlafaxine extended release tablets 9
Product Label Images
The following 11 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 13811-712 by Trigen Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
225MG 30CT - venlafaxine extended release tablets 10

This is a pharmaceutical product in tablet form coded as NDC 1381171530 Rconly 9. Each tablet contains 25mg of Venlafaxine in an extended-release formulation. The recommended dosage is once daily at 75mg strength. The product should be stored between 15-30°C. Controlled room temperature is recommended and excursions are permitted. A medication guide is enclosed with the product.*
225MG 90CT - venlafaxine extended release tablets 11

Venlafaxine Extended-Release Tablets, each containing 225 mg of venlafaxine in an extended-release formula, with one tablet recommended per day. Dosage information is available in the package insert. It should be stored at temperatures between 15-30°C and protected from moisture and humidity, with the attached Unit of Use Medication Guide provided each time it is dispensed. This medication is available only by prescription.*
37.5MG 30CT - venlafaxine extended release tablets 4

Each tablet contains 37.5mg venlafaxine in an extended-release formulation. The usual dosage is once daily and dosage information is available in the package insert. It should be stored at 25°C (I7°F) with excursions permitted at 10-15% 30°C (59°-86°F) and protected from moisture and humidity. These Venlafaxine Extended Release Tablets are available in a unit of use with 30 tablets in each. The manufacturer is Trigen Laboratories, LLC located in Edgewater, NJ 08607. A medication guide should be given to the patient when dispensing.*
37.5MG 90CT - venlafaxine extended release tablets 5

This appears to be the medication guide for a product containing Venlafaxine hydrochloride, an extended-release tablet prescribed for the treatment of depression. The recommended dosage is one tablet daily, and the medication should be stored at a temperature of 25°C (77°F). The package contains 90 tablets in a unit-of-use format.*
75MG30CT - venlafaxine extended release tablets 6

Each tablet contains 75mg of Venlafaxine in an extended-release formulation. Usual dosage is once daily, see package insert for more information. Store at controlled room temperature and protect from moisture and humidity. These tablets come in a unit of use medication guide and are marketed by Trigon Laboratories, LLC in Bridgewater, NJ. NDC 1381171330.*
150MG 30CT - venlafaxine extended release tablets 8

Each tablet of Venlafaxine contains 150 mg of the drug in an extended-release format. The standard dosage is one tablet per day, and the package insert contains detailed information on the same. The tablets should be stored at room temperature around 25°C, with slight excursions permitted. While handling the tablets, protect them from humidity and moisture. The medication guide should accompany the drug whenever the extended-release tablets are dispensed. The package contains 30 tablets, and the manufacturers are Trigen Laboratories LLC, located in Bridgewater, NJ 08807.*
150MG 90CT - venlafaxine extended release tablets 9

This is a medication guide for a package of 90 extended release tablets of Venlafaxine Hydrochloride, equivalent to 150mg dose. The usual dosage is once daily. The tablets are stored at room temperature. There are instructions for the authorized dispenser to control the administration of this medication. Each tablet is protected and meant for one-time use only.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.