FDA Label for Neuragen Pain Relief
View Indications, Usage & Precautions
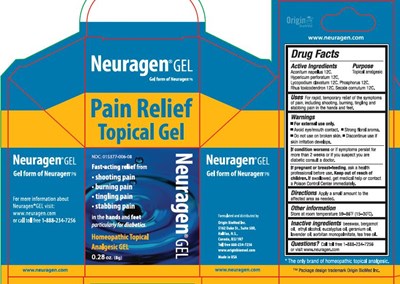
Neuragen Pain Relief Product Label
The following document was submitted to the FDA by the labeler of this product Origin Biomed Inc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Inactive Ingredient
bergamot oil, beeswax, ethyl alcohol, eucalyptus oil, geranium oil, lavender oil, sorbitan monopalmitate, tea tree oil
Dosage & Administration
Apply a small amount to the affected area as needed.
Warnings
- For external use only.
- Avoid eye/mouth contact
- Strong floral aroma
- Do not use on broken skin.
- Discontinue use if skin irritation develops
If condition worsens or if symptoms persist for more than 2 weeks or if you suspect you are diabetic consult a doctor.
Otc - Active Ingredient
Aconitum Napellus 12 C, Hypericum perforatum 12 C, Lycopodium clavatum 12 C, Phosphorus 12 C, Rhus toxicodendron 12 C, Secale cornutum 12 C
Otc - Purpose
Topical analgesic
Otc - Pregnancy Or Breast Feeding
If pregnant or breast feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Indications & Usage
For rapid, temporary relief of the symptoms of pain, including shooting, burning, tingling and stabbing pain in the hands and feet.
Storage And Handling
Store at room temperature 59-86 degrees F (15-30 degrees C)
Otc - Questions
Call toll free 1-888-234-7256 or visit www.neuragen.com
Package Label.Principal Display Panel
* Please review the disclaimer below.