Product Images Lansoprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Lansoprazole NDC 16590-891 by Stat Rx Usa Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label Image - LANSOPRAZOLE DR 15MG LABEL 890

This is a medication package containing Lansoprazole 15mg Delayed-Release capsules distributed by Sandoz Inc. The package was packaged and distributed from Gainesville, GA with a batch code of DC 1659089030. The manufacturer's product code is NOC 0781-2353-31 and the sample is labeled MD 1111712011.*
Label Image - LANSOPRAZOLE DR 30MG LABEL 891

This is a description of Lansoprazole capsules. The capsules are packaged and produced by Sandoz Inc in Princeton, NJ. They are delayed-release capsules containing 30mg of Lansoprazole. The lot number for the capsules is A235588 and the expiration date is 07/14 MD 11/17/2011. The NDC number is 16590-891-30.*
Figure - lansoprazole 02
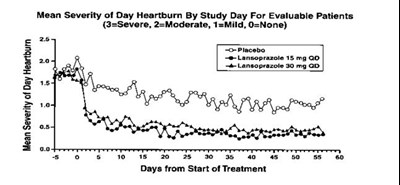
This is a graph showing the mean severity of day heartburn for evaluable patients over the course of treatment with a placebo, 15 mg Lansoprazole, and 30 mg Lansoprazole. The severity is rated on a scale of 1 to 3, with 3 being severe and 1 being mild. The x-axis shows the days from the start of treatment.*
Figure - lansoprazole 03
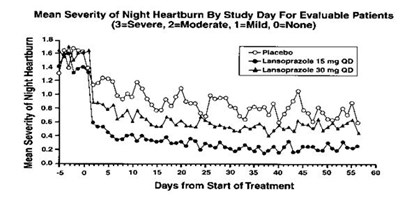
The text describes a graph showing the mean severity of night heartburn for evaluable patients over the days from the start of the treatment. The severity is measured on a scale from none to severe. However, there is no image of the graph available to analyze.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
