Product Images Budesonide Inhalation Suspension
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Budesonide Inhalation Suspension NDC 16714-020 by Northstar Rxllc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
0.5mg/2ml-pouch-label - 9bbd2475 b4f2 4776 b59e add17f17984b 09

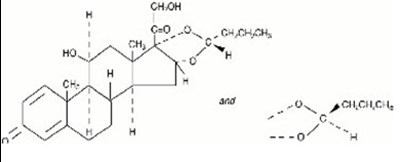
This is a description of a medication called "Budesonide Inhalation Suspension". It is packaged in an envelope containing five 2mL ampules. Each ampule includes a sterile suspension with 0.5mg of micronized budesonide with additional ingredients. The usual dosage can be found in the package insert, and the medication must be used in a jet nebulizer only. It should be stored at a controlled room temperature between 20°C to 25°C and should not be frozen. The medication must be used immediately once an ampule is opened, and the contents can be used within two weeks of opening. The medication should be kept away from the reach of children. The medication is manufactured by Cipla Ltd and distributed by Northstar Rx LLC in Memphis, TN 38141.*
1mg/2ml-pouch-label - 9bbd2475 b4f2 4776 b59e add17f17984b 11

This is a description of a medication called Budesonide Inhalation Suspension. The medication comes in single-dose ampules, containing a sterile suspension of 1 mg of micronized budesonide plus citric acid monohydrate, disodium edetate, polysorbate 80, sodium chloride, sodium citrate dihydrate, and water for injection. It is for inhalation use only and should be used in a jet nebulizer, not an ultrasonic nebulizer. The package insert should be consulted for the usual dosage, and the medication should be shaken before use. Once an ampule is opened, the contents should be used immediately, and the foil envelope should be kept until the last ampule is used. The medication should be stored at room temperature, protected from light, and out of reach of children. This medication is manufactured by Cipla Ltd. in India and is distributed by Northstar Rx LLC in Memphis, TN.*
1mg/2ml-carton-label - 9bbd2475 b4f2 4776 b59e add17f17984b 12

This is a description of a medication - Budesonide Inhalation Suspension for inhalation use only. It comes in a carton that contains single-dose ampules of 1mg/2 mL of Budesonide. The medication is from Northstar.*
label - 9bbd2475 b4f2 4776 b59e add17f17984b 13

This is a description of a medication called Budesonide Inhalation Suspension, which is for inhalation only. The medication comes in an envelope containing five 2 mL single-dose ampules, each delivering 0.25 mg of micronized budesonide plus other ingredients. The medication is meant to be used in a jet nebulizer and ampules need to be used immediately upon opening, and the patient should follow the package insert and the instructions from their doctor. It should be stored at 20°C to 25°C (88°F to 77°F), and the foil envelope should not be discarded until the last ampule is used. The drug should be stored in a place that is protected from light and out of the reach of children. The medication is manufactured by Cipla Ltd in India and is distributed by Northstar Rx LLC in Memphis, TN.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.