Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 01
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 02
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 03
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 04
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 05
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 06
- Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 07
Product Label Images
The following 7 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 16714-044 by Northstar Rx Llc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 02

This appears to be a drug interaction table showing different drugs and their possible interactions with each other. The drugs listed include ethanol, diazepam, alprazolam, indinavir, metoprolol, risperidone, imipramine, haloperidol, caffeine, and lithium. The table provides information on changes in PKFold values, recommended dosage adjustments, and clinical significances. Some drugs are listed as having no dose adjustment needed, while others are advised to be used with caution or avoided entirely. Overall, this provides important information for healthcare professionals to consider when managing a patient's medication regimen.*
Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 03
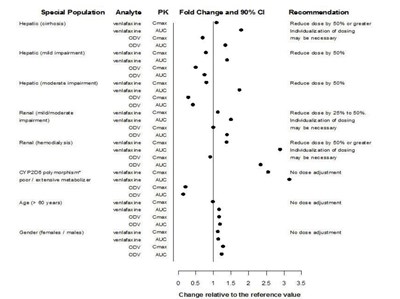
This appears to be a table or chart with information related to the impact of certain factors on the pharmacokinetics (PK) of the drug venlafaxine. The factors include special populations with liver or renal impairment, CYP2D8 polymorphism, age, gender, and the analyte of interest. The table provides recommendations for dose adjustments based on fold change and 90% confidence interval. It appears to be a guide to help health professionals individualize dosing for patients in different categories.*
Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 05

This is a description of a medication called Venlafaxine Hydrochloride Extended-Release Capsules, USP. The medication comes in a bottle containing 90 capsules and should be stored at a temperature between 21 to 25 degrees Celsius. The packaging also provides the NDC number (16714-043-01) and some dosage information, but includes some errors.*
Venlafaxine hydrochloride extended-release capsules, USP - 310c558f 5135 4ddd b0bd f993c0ea8d62 06

This is a description of a medication with the NDC code of 16714-044-01. The medication is in the form of extended-release capsules which contain 75mg of Venlafaxine equivalent to Venlafarie. The capsules also have Hydrochloride e pessang mamsn. The extended-release capsules are to be kept out of reach of children and pets. A medication guide is provided separately. The manufacturer of the drug is Norhetar Rx LLC in Meghi, TN and was manufactured by Cadia Healircare Ltd in Ahmedabad, India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


