FDA Label for Ezetimibe And Simvastatin
View Indications, Usage & Precautions
- 1 INDICATIONS AND USAGE
- 1.1 PRIMARY HYPERLIPIDEMIA
- 1.2 HOMOZYGOUS FAMILIAL HYPERCHOLESTEROLEMIA (HOFH)
- 1.3 LIMITATIONS OF USE
- 2.1 RECOMMENDED DOSING
- 2.2 RESTRICTED DOSING FOR 10/80 MG
- 2.3 COADMINISTRATION WITH OTHER DRUGS
- 2.4 PATIENTS WITH HOMOZYGOUS FAMILIAL HYPERCHOLESTEROLEMIA
- 2.5 PATIENTS WITH RENAL IMPAIRMENT/CHRONIC KIDNEY DISEASE
- 2.6 GERIATRIC PATIENTS
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 MYOPATHY/RHABDOMYOLYSIS
- 5.3 LIVER ENZYMES
- 5.4 ENDOCRINE FUNCTION
- 6 ADVERSE REACTIONS
- 6.1 CLINICAL TRIALS EXPERIENCE
- 6.2 POSTMARKETING EXPERIENCE
- 7 DRUG INTERACTIONS
- 7.1 STRONG CYP3A4 INHIBITORS, CYCLOSPORINE, OR DANAZOL
- 7.2 LIPID-LOWERING DRUGS THAT CAN CAUSE MYOPATHY WHEN GIVEN ALONE
- 7.3 AMIODARONE, DRONEDARONE, RANOLAZINE, OR CALCIUM CHANNEL BLOCKERS
- 7.4 NIACIN
- 7.5 CHOLESTYRAMINE
- 7.6 DIGOXIN
- 7.7 FENOFIBRATES (E.G., FENOFIBRATE AND FENOFIBRIC ACID)
- 7.8 COUMARIN ANTICOAGULANTS
- 7.9 COLCHICINE
- 7.10 DAPTOMYCIN
- 8.1 PREGNANCY
- 8.3 NURSING MOTHERS
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 8.6 RENAL IMPAIRMENT
- 8.7 HEPATIC IMPAIRMENT
- 8.8 CHINESE PATIENTS
- 10 OVERDOSAGE
- 11 DESCRIPTION
- 12.1 MECHANISM OF ACTION
- 12.2 PHARMACODYNAMICS
- 12.3 PHARMACOKINETICS
- 13.1 CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
- 13.2 ANIMAL TOXICOLOGY AND/OR PHARMACOLOGY
- 14.1 PRIMARY HYPERLIPIDEMIA
- 14.2 HOMOZYGOUS FAMILIAL HYPERCHOLESTEROLEMIA (HOFH)
- 14.3 CHRONIC KIDNEY DISEASE (CKD)
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- 17.1 MUSCLE PAIN
- 17.2 LIVER ENZYMES
- 17.3 PREGNANCY
- 17.4 BREASTFEEDING
- OTHER
- SPL PATIENT PACKAGE INSERT
- PRINCIPAL DISPLAY PANEL - 10 MG/10 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 10 MG/20 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 10 MG/40 MG TABLET BOTTLE LABEL
- PRINCIPAL DISPLAY PANEL - 10 MG/80 MG TABLET BOTTLE LABEL
Ezetimibe And Simvastatin Product Label
The following document was submitted to the FDA by the labeler of this product Northstar Rx Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
1 Indications And Usage
1.1 Primary Hyperlipidemia
1.2 Homozygous Familial Hypercholesterolemia (Hofh)
1.3 Limitations Of Use
2.1 Recommended Dosing
2.2 Restricted Dosing For 10/80 Mg
2.3 Coadministration With Other Drugs
Patients taking Verapamil, Diltiazem, or Dronedarone
• The dose of ezetimibe and simvastatin tablets should not exceed 10/10 mg/day [see Warnings and Precautions (5.1), Drug Interactions (7.3), and Clinical Pharmacology (12.3)].• The dose of ezetimibe and simvastatin tablets should not exceed 10/20 mg/day [see Warnings and Precautions (5.1), Drug Interactions (7.3), and Clinical Pharmacology (12.3)].• Dosing of ezetimibe and simvastatin tablets should occur either greater than or equal to 2 hours before or greater than or equal to 4 hours after administration of a bile acid sequestrant [see Drug Interactions (7.5)].
Patients taking Amiodarone, Amlodipine or Ranolazine
Patients taking Bile Acid Sequestrants
2.4 Patients With Homozygous Familial Hypercholesterolemia
2.5 Patients With Renal Impairment/Chronic Kidney Disease
2.6 Geriatric Patients
3 Dosage Forms And Strengths
• Ezetimibe and Simvastatin Tablets 10 mg/10 mg are white to off-white, capsule shaped, uncoated tablets debossed with ‘G’ on one side and ‘321’ on the other side.• Ezetimibe and Simvastatin Tablets 10 mg/20 mg are white to off-white, capsule shaped, uncoated tablets• Ezetimibe and Simvastatin Tablets 10 mg/40 mg are white to off-white, capsule shaped, uncoated tablets debossed with ‘G’ on one side and ‘323’ on the other side.• Ezetimibe and Simvastatin Tablets 10 mg/80 mg are white to off-white, capsule shaped, uncoated tablets debossed with ‘G’ on one side and ‘324’ on the other side.
4 Contraindications
Ezetimibe and simvastatin tablets are contraindicated in the following conditions:
• Concomitant administration of strong CYP3A4 inhibitors (e.g., itraconazole, ketoconazole, posaconazole, voriconazole, HIV protease inhibitors, boceprevir, telaprevir, erythromycin, clarithromycin, telithromycin, nefazodone, and cobicistat-containing products) [see Warnings and Precautions (5.1)].• Concomitant administration of gemfibrozil, cyclosporine, or danazol [see Warnings and Precautions (5.1)].• Hypersensitivity to any component of this medication [see Adverse Reactions (6.2)].• Active liver disease or unexplained persistent elevations in hepatic transaminase levels [see Warnings and Precautions (5.3)].• Women who are pregnant or may become pregnant. Serum cholesterol and triglycerides increase during normal pregnancy, and cholesterol or cholesterol derivatives are essential for fetal development. Because HMG-CoA reductase inhibitors (statins), such as simvastatin, decrease cholesterol synthesis and possibly the synthesis of other biologically active substances derived from cholesterol, ezetimibe and simvastatin may cause fetal harm when administered to a pregnant woman. Atherosclerosis is a chronic process and the discontinuation of lipid-lowering drugs during pregnancy should have little impact on the outcome of long-term therapy of primary hypercholesterolemia. There are no adequate and well-controlled studies of ezetimibe and simvastatin use during pregnancy; however, in rare reports congenital anomalies were observed following intrauterine exposure to statins. In rat and rabbit animal reproduction studies, simvastatin revealed no evidence of teratogenicity. Ezetimibe and simvastatin should be administered to women of childbearing age only when such patients are highly unlikely to conceive. If the patient becomes pregnant while taking this drug, ezetimibe and simvastatin should be discontinued immediately and the patient should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)].• Nursing mothers. It is not known whether simvastatin is excreted into human milk; however, a small amount of another drug in this class does pass into breast milk. Because statins have the potential for serious adverse reactions in nursing infants, women who require ezetimibe and simvastatin treatment should not breastfeed their infants [see Use in Specific Populations (8.3)].
5.1 Myopathy/Rhabdomyolysis
The benefits of the combined use of ezetimibe and simvastatin tablets with the following drugs should be carefully weighed against the potential risks of combinations: other lipid-lowering drugs (fenofibrates or, for patients with HoFH, lomitapide), amiodarone, dronedarone, verapamil, diltiazem, amlodipine, or ranolazine [see Dosage and Administration (2.4), Drug Interactions (7.3)].
Interacting Agents | Prescribing Recommendations |
Gemfibrozil Cyclosporine Danazol | Contraindicated with ezetimibe and simvastatin tablets |
Niacin (≥1 g/day) | For Chinese patients, not recommended with ezetimibe and simvastatin tablets |
Verapamil Diltiazem Dronedarone | Do not exceed 10/10 mg ezetimibe and simvastatin tablets daily |
Amiodarone Amlodipine Ranolazine | Do not exceed 10/20 mg ezetimibe and simvastatin tablets daily |
Lomitapide | For patients with HoFH, do not exceed 10/20 mg ezetimibe and simvastatin tablets daily* |
Daptomycin | Temporarily suspend ezetimibe and simvastatin |
Grapefruit juice | Avoid grapefruit juice |
* For patients with HoFH who have been taking 80 mg simvastatin chronically (e.g., for 12 months or more) without evidence of muscle toxicity, do not exceed 10/40 mg ezetimibe and simvastatin tablets when taking lomitapide. | |
5.3 Liver Enzymes
5.4 Endocrine Function
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the label:
6.1 Clinical Trials Experience
Ezetimibe and Simvastatin Tablets
• Increased ALT (0.9%)• Myalgia (0.6%)• Increased AST (0.4%)• Back pain (0.4%)
Body System/Organ Class Adverse Reaction | Placebo (%) n=371 | Ezetimibe 10 mg (%) n=302 | Simvastatin† (%) n=1234 | Ezetimibe and Simvastatin Tablets† (%) n=1420 |
Body as a whole – general disorders | ||||
Headache | 5.4 | 6 | 5.9 | 5.8 |
Gastrointestinal system disorders | ||||
Diarrhea | 2.2 | 5 | 3.7 | 2.8 |
Infections and infestations | ||||
Influenza | 0.8 | 1 | 1.9 | 2.3 |
Upper respiratory tract infection | 2.7 | 5 | 5 | 3.6 |
Musculoskeletal and connective tissue disorders | ||||
Myalgia | 2.4 | 2.3 | 2.6 | 3.6 |
Pain in extremity | 1.3 | 3 | 2 | 2.3 |
* Includes two placebo-controlled combination studies in which the active ingredients equivalent to ezetimibe and simvastatin tablets were coadministered and two placebo-controlled studies in which ezetimibe and simvastatin tablets was administered. † All doses. | ||||
Study of Heart and Renal Protection
Ezetimibe
Simvastatin
Laboratory Tests
Marked persistent increases of hepatic serum transaminases have been noted [see Warnings and Precautions (5.3)]. Elevated alkaline phosphatase and γ-glutamyl transpeptidase have been reported. About 5% of patients taking simvastatin had elevations of CK levels of 3 or more times the normal value on one or more occasions. This was attributable to the noncardiac fraction of CK [see Warnings and Precautions (5.1)].
6.2 Postmarketing Experience
There have been rare postmarketing reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. These cognitive issues have been reported for all statins. The reports are generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).
7 Drug Interactions
[See Clinical Pharmacology (12.3)].
Ezetimibe and simvastatin tablets
7.1 Strong Cyp3a4 Inhibitors, Cyclosporine, Or Danazol
Cyclosporine or Danazol: The risk of myopathy, including rhabdomyolysis is increased by concomitant administration of cyclosporine or danazol. Therefore, concomitant use of these drugs is contraindicated [see Contraindications (4), Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
7.2 Lipid-Lowering Drugs That Can Cause Myopathy When Given Alone
Fenofibrates (e.g., fenofibrate and fenofibric acid): Caution should be used when prescribing with ezetimibe and simvastatin tablets [see Warnings and Precautions (5.1) and Drug Interactions (7.7)].
7.3 Amiodarone, Dronedarone, Ranolazine, Or Calcium Channel Blockers
The risk of myopathy, including rhabdomyolysis, is increased by concomitant administration of amiodarone, dronedarone, ranolazine, or calcium channel blockers such as verapamil, diltiazem or amlodipine [see Dosage and Administration (2.3) and Warnings and Precautions (5.1) and Table 6 in Clinical Pharmacology (12.3)].
7.4 Niacin
Cases of myopathy/rhabdomyolysis have been observed with simvastatin coadministered with lipid-modifying doses (≥1 g/day niacin) of niacin-containing products. The risk of myopathy is greater in Chinese patients. In a clinical trial (median follow-up 3.9 years) involving patients at high risk of cardiovascular disease and with well-controlled LDL-C levels on simvastatin 40 mg/day with or without ezetimibe 10 mg/day, there was no incremental benefit on cardiovascular outcomes with the addition of lipid-modifying doses (≥1 g/day) of niacin. Coadministration of ezetimibe and simvastatin tablets with lipid-modifying doses (≥1 g/day) of niacin is not recommended in Chinese patients. It is unknown if this risk applies to other Asian patients [see Warnings and Precautions (5.1) and Use in Specific Populations (8.8)].
7.5 Cholestyramine
Concomitant cholestyramine administration decreased the mean AUC of total ezetimibe approximately 55%. The incremental LDL-C reduction due to adding ezetimibe and simvastatin tablets to cholestyramine may be reduced by this interaction.
7.6 Digoxin
In one study, concomitant administration of digoxin with simvastatin resulted in a slight elevation in plasma digoxin concentrations. Patients taking digoxin should be monitored appropriately when ezetimibe and simvastatin tablets are initiated.
7.7 Fenofibrates (E.G., Fenofibrate And Fenofibric Acid)
Fenofibrates may increase cholesterol excretion into the bile, leading to cholelithiasis. In a preclinical study in dogs, ezetimibe increased cholesterol in the gallbladder bile [see Animal Toxicology and/or Pharmacology (13.2)]. If cholelithiasis is suspected in a patient receiving ezetimibe and simvastatin tablets and a fenofibrate, gallbladder studies are indicated and alternative lipid-lowering therapy should be considered [see the product labeling for fenofibrate and fenofibric acid].
7.8 Coumarin Anticoagulants
The effect of ezetimibe and simvastatin tablets on the prothrombin time has not been studied.
7.9 Colchicine
Cases of myopathy, including rhabdomyolysis, have been reported with simvastatin coadministered with colchicine, and caution should be exercised when prescribing ezetimibe and simvastatin tablets with colchicine.
7.10 Daptomycin
Cases of rhabdomyolysis have been reported with ezetimibe and simvastatin administered with daptomycin. Both ezetimibe and simvastatin and daptomycin can cause myopathy and rhabdomyolysis when given alone and the risk of myopathy and rhabdomyolysis may be increased by coadministration. Temporarily suspend ezetimibe and simvastatin in patients taking daptomycin [see Warnings and Precautions (5.1)].
8.1 Pregnancy
Pregnancy Category X.
Ezetimibe and simvastatin tablets
Ezetimibe
Simvastatin
Simvastatin was not teratogenic in rats or rabbits at doses (25, 10 mg/kg/day, respectively) that resulted in 3 times the human exposure based on mg/m2 surface area. However, in studies with another structurally-related statin, skeletal malformations were observed in rats and mice.
There are rare reports of congenital anomalies following intrauterine exposure to statins. In a review1 of approximately 100 prospectively followed pregnancies in women exposed to simvastatin or another structurally-related statin, the incidences of congenital anomalies, spontaneous abortions and fetal deaths/stillbirths did not exceed what would be expected in the general population. The number of cases is adequate only to exclude a 3- to 4-fold increase in congenital anomalies over the background incidence. In 89% of the prospectively followed pregnancies, drug treatment was initiated prior to pregnancy and was discontinued at some point in the first trimester when pregnancy was identified.
8.3 Nursing Mothers
8.4 Pediatric Use
The effects of ezetimibe coadministered with simvastatin (n=126) compared to simvastatin monotherapy (n=122) have been evaluated in adolescent boys and girls with heterozygous familial hypercholesterolemia (HeFH). In a multicenter, double-blind, controlled study followed by an open-label phase, 142 boys and 106 postmenarchal girls, 10 to 17 years of age (mean age 14.2 years, 43% females, 82% Caucasians, 4% Asian, 2% Blacks, 13% multiracial) with HeFH were randomized to receive either ezetimibe coadministered with simvastatin or simvastatin monotherapy. Inclusion in the study required 1) a baseline LDL-C level between 160 and 400 mg/dL and 2) a medical history and clinical presentation consistent with HeFH. The mean baseline LDL-C value was 225 mg/dL (range: 161 to 351 mg/dL) in the ezetimibe coadministered with simvastatin group compared to 219 mg/dL (range: 149 to 336 mg/dL) in the simvastatin monotherapy group. The patients received coadministered ezetimibe and simvastatin (10 mg, 20 mg, or 40 mg) or simvastatin monotherapy (10 mg, 20 mg, or 40 mg) for 6 weeks, coadministered ezetimibe and 40 mg simvastatin or 40 mg simvastatin monotherapy for the next 27 weeks, and open-label coadministered ezetimibe and simvastatin (10 mg, 20 mg, or 40 mg) for 20 weeks thereafter.
The results of the study at Week 6 are summarized in Table 3. Results at Week 33 were consistent with those at Week 6.
| Total-C | LDL-C | Apo B | Non-HDL-C | TG* | HDL-C | |
|---|---|---|---|---|---|---|
Mean percent difference between treatment groups | -12% | -15% | -12% | -14% | -2% | +0.1% |
95% Confidence Interval | (-15%, -9%) | (-18%, -12%) | (-15%, -9%) | (-17%, -11%) | (-9, +4) | (-3, +3) |
* For triglycerides, median % change from baseline. | ||||||
From the start of the trial to the end of Week 33, discontinuations due to an adverse reaction occurred in 7 (6%) patients in the ezetimibe coadministered with simvastatin group and in 2 (2%) patients in the simvastatin monotherapy group.
During the trial, hepatic transaminase elevations (two consecutive measurements for ALT and/or AST ≥3 X ULN) occurred in four (3%) individuals in the ezetimibe coadministered with simvastatin group and in two (2%) individuals in the simvastatin monotherapy group. Elevations of CPK (≥10 X ULN) occurred in two (2%) individuals in the ezetimibe coadministered with simvastatin group and in zero individuals in the simvastatin monotherapy group.
In this limited controlled study, there was no significant effect on growth or sexual maturation in the adolescent boys or girls, or on menstrual cycle length in girls.
Coadministration of ezetimibe with simvastatin at doses greater than 40 mg/day has not been studied in adolescents. Also, ezetimibe and simvastatin tablets have not been studied in patients younger than 10 years of age or in pre-menarchal girls.
Ezetimibe
Based on total ezetimibe (ezetimibe + ezetimibe-glucuronide) there are no pharmacokinetic differences between adolescents and adults. Pharmacokinetic data in the pediatric population <10 years of age are not available.
Simvastatin
The pharmacokinetics of simvastatin has not been studied in the pediatric population.
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
8.8 Chinese Patients
In a clinical trial in which patients at high risk of cardiovascular disease were treated with simvastatin 40 mg/day (median follow-up 3.9 years), the incidence of myopathy was approximately 0.05% for non- Chinese patients (n=7367) compared with 0.24% for Chinese patients (n=5468). The incidence of myopathy for Chinese patients on simvastatin 40 mg/day or ezetimibe and simvastatin 10/40 mg/day coadministered with extended-release niacin 2 g/day was 1.24%.
Chinese patients may be at higher risk for myopathy, monitor patients appropriately. Coadministration of ezetimibe and simvastatin tablets with lipid-modifying doses (≥1 g/day niacin) of niacin-containing products is not recommended in Chinese patients [see Warnings and Precautions (5.1), Drug Interactions (7.4)].
10 Overdosage
Ezetimibe and simvastatin tablets
Ezetimibe
Simvastatin
11 Description
Ezetimibe and Simvastatin Tablets contain ezetimibe, a selective inhibitor of intestinal cholesterol and related phytosterol absorption, and simvastatin, an HMG-CoA reductase inhibitor.
The chemical name of ezetimibe is 1-(4-fluorophenyl)-3(R)-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4(S)-(4-hydroxyphenyl)-2-azetidinone. The molecular formula is C24H21F2NO3 and its molecular weight is 409.44 g/mol.
Ezetimibe is a white, crystalline powder that is freely soluble in ethanol, methanol and acetone and practically insoluble in water. Its structural formula is:
Simvastatin, an inactive lactone, is hydrolyzed to the corresponding β-hydroxyacid form, which is an inhibitor of HMG-CoA reductase. Simvastatin is butanoic acid, 2,2-dimethyl-,1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)-ethyl]-1-naphthalenyl ester, [1S-[1α,3α,7α,8α(2S*,4S*),-8aβ]]. The molecular formula of simvastatin is C25H38O5 and its molecular weight is 418.57 g/mol.
Simvastatin is a white to off-white, nonhygroscopic powder that is freely soluble in chloroform, methanol and alcohol, sparingly soluble in propylene glycol, very slightly soluble in hexane and practically insoluble in water. Its structural formula is:
Ezetimibe and Simvastatin Tablets are available for oral use as tablets containing 10 mg of ezetimibe, and 10 mg of simvastatin (Ezetimibe and Simvastatin Tablets 10 mg/10 mg), 20 mg of simvastatin (Ezetimibe and Simvastatin Tablets 10 mg/20 mg), 40 mg of simvastatin (Ezetimibe and Simvastatin Tablets 10 mg/40 mg), or 80 mg of simvastatin (Ezetimibe and Simvastatin Tablets 10 mg/80 mg). Each tablet contains the following inactive ingredients: butylated hydroxyanisole, citric acid monohydrate, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, propyl gallate, and sodium lauryl sulfate.
12.1 Mechanism Of Action
Ezetimibe and Simvastatin Tablets
Simvastatin
Ezetimibe
12.2 Pharmacodynamics
12.3 Pharmacokinetics
Simvastatin
Simvastatin
Simvastatin
Ezetimibe
Absorption
Ezetimibe
Simvastatin
Effect of Food on Oral Absorption
Ezetimibe
Distribution
Ezetimibe
Simvastatin
Metabolism and Excretion
Ezetimibe
Simvastatin
Specific Populations
Geriatric Patients
Ezetimibe
Pediatric Patients: [See Use in Specific Populations (8.4).]
Gender
Ezetimibe
Race
Ezetimibe
Hepatic Impairment
Ezetimibe
Renal Impairment
Ezetimibe
Drug Interactions [See also Drug Interactions (7).]
Table 4: Effect of Coadministered Drugs on Total Ezetimibe
Coadministered Drug and Dosing Regimen | Total Ezetimibe* | |
Change in AUC | Change in Cmax | |
Cyclosporine-stable dose required (75 to 150 mg BID)†,‡ | ↑240% | ↑290% |
Fenofibrate, 200 mg QD, 14 days‡ | ↑48% | ↑64% |
Gemfibrozil, 600 mg BID, 7 days‡ | ↑64% | ↑91% |
Cholestyramine, 4 g BID, 14 days‡ | ↓55% | ↓4% |
Aluminum & magnesium hydroxide combination antacid, single dose§ | ↓4% | ↓30% |
Cimetidine, 400 mg BID, 7 days | ↑6% | ↑22% |
Glipizide, 10 mg, single dose | ↑4% | ↓8% |
Statins | ||
↑9% | ↑3% | |
↑7% | ↑23% | |
↓2% | ↑12% | |
↑13% | ↑18% | |
↓19% | ↑7% | |
| Coadministered Drug and its Dosage Regimen | Ezetimibe Dosage Regimen | Change in AUC of Coadministered Drug | Change in Cmax of Coadministered Drug |
|---|---|---|---|
Warfarin, 25 mg single dose on Day 7 | 10 mg QD, 11 days | ↓2% (R-warfarin) ↓4% (S-warfarin) | ↑3% (R-warfarin) ↑1% (S-warfarin) |
Digoxin, 0.5 mg single dose | 10 mg QD, 8 days | ↑2% | ↓7% |
Gemfibrozil, 600 mg BID, 7 days* | 10 mg QD, 7 days | ↓1% | ↓11% |
Ethinyl estradiol & Levonorgestrel, QD, 21 days | 10 mg QD, Days 8 to 14 of 21 day oral contraceptive cycle | Ethinyl estradiol 0% Levonorgestrel 0% | Ethinyl estradiol ↓9% Levonorgestrel ↓5% |
Glipizide, 10 mg on Days 1 and 9 | 10 mg QD, Days 2 to 9 | ↓3% | ↓5% |
Fenofibrate, 200 mg QD, 14 days* | 10 mg QD, 14 days | ↑11% | ↑7% |
Cyclosporine, 100 mg single dose Day 7* | 20 mg QD, 8 days | ↑15% | ↑10% |
Statins | |||
10 mg QD, 7 days | ↑19% | ↑3% | |
10 mg QD, 14 days | ↓20% | ↓24% | |
10 mg QD, 14 days | ↓4% | ↑7% | |
10 mg QD, 14 days | ↑19% | ↑17% | |
10 mg QD, 14 days | ↓39% | ↓27% | |
* See 7. Drug Interactions. | |||
Simvastatin
| Coadministered Drug or Grapefruit Juice | Dosing of Coadministered Drug or Grapefruit Juice | Dosing of Simvastatin | Geometric Mean Ratio (Ratio* with / without coadministered drug) No Effect = 1 | ||
|---|---|---|---|---|---|
| AUC | Cmax | ||||
Contraindicated with Ezetimibe and Simvastatin Tablets [see Contraindications (4) and Warnings and Precautions (5.1)] | |||||
Telithromycin† | 200 mg QD for 4 days | 80 mg | simvastatin acid‡ simvastatin | 12 8.9 | 15 5.3 |
Nelfinavir† | 1250 mg BID for 14 days | 20 mg QD for 28 days | simvastatin acid‡ simvastatin |
|
|
Itraconazole† | 200 mg QD for 4 days | 80 mg | simvastatin acid‡ simvastatin | 13.1 13.1 | |
Posaconazole | 100 mg (oral suspension) QD for 13 days | 40 mg
| simvastatin acid‡ simvastatin | 7.3 10.3 8.5 10.6 | 9.2 9.4 9.5 11.4 |
Gemfibrozil | 600 mg BID for 3 days | 40 mg | simvastatin acid‡ simvastatin | 2.85 1.35 | 2.18 0.91 |
Avoid grapefruit juice with Ezetimibe and Simvastatin Tablets [see Warnings and Precautions (5.1)] | |||||
Grapefruit Juice§ (high dose) | 200 mL of double-strength TID¶ | 60 mg single dose | simvastatin acid simvastatin | 7 16 | |
Grapefruit Juice§ (low dose) | 8 oz (about 237 mL) of single-strength# | 20 mg single dose | simvastatin acid simvastatin | 1.3 1.9 | |
Avoid taking with >10/10 mg Ezetimibe and Simvastatin Tablets, based on clinical and/or postmarketing simvastatin experience [see Warnings and Precautions (5.1)] | |||||
Verapamil SR | 240 mg QD Days 1 to 7 then 240 mg BID on Days 8 to 10 | 80 mg on Day 10 | simvastatin acid simvastatin | 2.3 2.5 | 2.4 2.1 |
Diltiazem | 120 mg BID for 10 days | 80 mg on Day 10 | simvastatin acid simvastatin | 2.69 3.10 | 2.69 2.88 |
Diltiazem | 120 mg BID for 14 days | 20 mg on Day 14 | simvastatin | 4.6 | 3.6 |
Dronedarone | 400 mg BID for 14 days | 40 mg QD for 14 days | simvastatin acid simvastatin | 1.96 3.90 | 2.14 3.75 |
Avoid taking with >10/20 mg Ezetimibe and Simvastatin Tablets, based on clinical and/or postmarketing simvastatin experience [see Warnings and Precautions (5.1)] | |||||
Amiodarone | 400 mg QD for 3 days | 40 mg on Day 3 | simvastatin acid simvastatin | 1.75 1.76 | 1.72 1.79 |
Amlodipine | 10 mg QD for 10 days | 80 mg on Day 10 | simvastatin acid simvastatin | 1.58 1.77 | 1.56 1.47 |
Ranolazine SR | 1000 mg BID for 7 days | 80 mg on Day 1 and Days 6 to 9 | simvastatin acid simvastatin | 2.26 1.86 | 2.28 1.75 |
Avoid taking with >10/20 mg Ezetimibe and Simvastatin Tablets (or 10/40 mg for patients who have previously taken 80 mg simvastatin chronically, e.g., for 12 months or more, without evidence of muscle toxicity), based on clinical experience | |||||
Lomitapide | 60 mg QD for 7 days | 40 mg single dose | simvastatin acid | 1.7 | 1.6 |
simvastatin | 2 | 2 | |||
Lomitapide | 10 mg QD for 7 days | 20 mg single dose | simvastatin acid | 1.4 | 1.4 |
simvastatin | 1.6 | 1.7 | |||
No dosing adjustments required for the following: | |||||
Fenofibrate | 160 mg QD for 14 days | 80 mg QD on Days 8 to 14 | simvastatin acid simvastatin | 0.64 0.89 | 0.89 0.83 |
Propranolol | 80 mg single dose | 80 mg single dose | total inhibitor | 0.79 | ↓ from 33.6 to 21.1 ng∙eq/mL |
* Results based on a chemical assay except results with propranolol as indicated. | |||||
13.1 Carcinogenesis, Mutagenesis, Impairment Of Fertility
Ezetimibe and Simvastatin Tablets
13.2 Animal Toxicology And/Or Pharmacology
CNS Toxicity
14.1 Primary Hyperlipidemia
Ezetimibe and Simvastatin Tablets
Table 7: Response to Ezetimibe and Simvastatin Tablets in Patients with Primary Hyperlipidemia (Mean* % Change from Untreated Baseline†)
| Treatment (Daily Dose) | N | Total-C | LDL-C | Apo B | HDL-C | TG* | Non-HDL-C |
|---|---|---|---|---|---|---|---|
Pooled data (All Ezetimibe and Simvastatin Tablets doses)‡ | 609 | -38 | -53 | -42 | +7 | -24 | -49 |
Pooled data (All simvastatin doses)‡ | 622 | -28 | -39 | -32 | +7 | -21 | -36 |
Ezetimibe 10 mg | 149 | -13 | -19 | -15 | +5 | -11 | -18 |
Placebo | 148 | -1 | -2 | 0 | 0 | -2 | -2 |
Ezetimibe and Simvastatin Tablets by dose | |||||||
10/10 | 152 | -31 | -45 | -35 | +8 | -23 | -41 |
10/20 | 156 | -36 | -52 | -41 | +10 | -24 | -47 |
10/40 | 147 | -39 | -55 | -44 | +6 | -23 | -51 |
10/80 | 154 | -43 | -60 | -49 | +6 | -31 | -56 |
Simvastatin by dose | |||||||
10 mg | 158 | -23 | -33 | -26 | +5 | -17 | -30 |
20 mg | 150 | -24 | -34 | -28 | +7 | -18 | -32 |
40 mg | 156 | -29 | -41 | -33 | +8 | -21 | -38 |
80 mg | 158 | -35 | -49 | -39 | +7 | -27 | -45 |
* For triglycerides, median % change from baseline. † Baseline - on no lipid-lowering drug. ‡ Ezetimibe and simvastatin tablets doses pooled (10/10 to 10/80) significantly reduced total-C, LDL-C, Apo B, TG, and non-HDL-C compared to simvastatin and significantly increased HDL-C compared to placebo. | |||||||
In a multicenter, double-blind, controlled, 23-week study, 710 patients with known CHD or CHD risk equivalents, as defined by the NCEP ATP III guidelines, and an LDL-C ≥130 mg/dL were randomized to one of four treatment groups: coadministered ezetimibe and simvastatin equivalent to ezetimibe and simvastatin tablets (10/10, 10/20, and 10/40) or simvastatin 20 mg. Patients not reaching an LDL-C <100 mg/dL had their simvastatin dose titrated at 6-week intervals to a maximal dose of 80 mg.
At Week 5, the LDL-C reductions with ezetimibe and simvastatin tablets 10/10, 10/20, or 10/40 were significantly larger
than with simvastatin 20 mg (see Table 8).
| Simvastatin 20 mg | Ezetimibe and Simvastatin Tablets 10/10 | Ezetimibe and Simvastatin Tablets 10/20 | Ezetimibe and Simvastatin Tablets 10/40 | |
|---|---|---|---|---|
N | 253 | 251 | 109 | 97 |
Mean baseline LDL-C | 174 | 165 | 167 | 171 |
Percent change LDL-C | -38 | -47 | -53 | -59 |
Table 9: Response to Ezetimibe and Simvastatin Tablets and Atorvastatin in Patients with Primary Hyperlipidemia (Mean* % Change from Untreated Baseline†)
| Treatment (Daily Dose) | N | Total-C‡ | LDL-C‡ | Apo B‡ | HDL-C | TG* | Non-HDL-C‡ |
|---|---|---|---|---|---|---|---|
Ezetimibe and Simvastatin Tablets by dose | |||||||
10/10 | 230 | -34§ | -47§ | -37§ | +8 | -26 | -43§ |
10/20 | 233 | -37§ | -51§ | -40§ | +7 | -25 | -46§ |
10/40 | 236 | -41§ | -57§ | -46§ | +9§ | -27 | -52§ |
10/80 | 224 | -43§ | -59§ | -48§ | +8§ | -31 | -54§ |
Atorvastatin by dose | |||||||
10 mg | 235 | -27 | -36 | -31 | +7 | -21 | -34 |
20 mg | 230 | -32 | -44 | -37 | +5 | -25 | -41 |
40 mg | 232 | -36 | -48 | -40 | +4 | -24 | -45 |
80 mg | 230 | -40 | -53 | -44 | +1 | -32 | -50 |
* For triglycerides, median % change from baseline. † Baseline - on no lipid-lowering drug. ‡ Ezetimibe and simvastatin tablets doses pooled (10/10 to 10/80) provided significantly greater reductions in total-C, LDL-C, Apo B, and non-HDL-C compared to atorvastatin doses pooled (10 to 80). § p<0.05 for difference with atorvastatin at equal mg doses of the simvastatin component. In a multicenter, double-blind, 24-week, forced-titration study, 788 patients with primary hyperlipidemia, who had not met their NCEP ATP III target LDL-C goal, were randomized to receive coadministered ezetimibe and simvastatin equivalent to ezetimibe and simvastatin tablets (10/10 and 10/20) or atorvastatin 10 mg. For all three treatment groups, the dose of the statin was titrated at 6-week intervals to 80 mg. At each pre-specified dose comparison, ezetimibe and simvastatin tablets lowered LDL-C to a greater degree than atorvastatin (see Table 10). | |||||||
Table 10: Response to Ezetimibe and Simvastatin Tablets and Atorvastatin in Patients with Primary Hyperlipidemia (Mean* % Change from Untreated Baseline†)
| Treatment | N | Total-C | LDL-C | Apo B | HDL-C | TG* | Non-HDL-C |
|---|---|---|---|---|---|---|---|
Week 6 | |||||||
Atorvastatin 10 mg‡ | 262 | -28 | -37 | -32 | +5 | -23 | -35 |
Ezetimibe and Simvastatin Tablets 10/10§ | 263 | -34¶ | -46¶ | -38¶ | +8¶ | -26 | -43¶ |
Ezetimibe and Simvastatin Tablets 10/20# | 263 | -36¶ | -50¶ | -41¶ | +10¶ | -25 | -46¶ |
Week 12 | |||||||
Atorvastatin 20 mg | 246 | -33 | -44 | -38 | +7 | -28 | -42 |
Ezetimibe and Simvastatin Tablets 10/20 | 250 | -37¶ | -50¶ | -41¶ | +9 | -28 | -46¶ |
Ezetimibe and Simvastatin Tablets 10/40 | 252 | -39¶ | -54¶ | -45¶ | +12¶ | -31 | -50¶ |
Week 18 | |||||||
Atorvastatin 40 mg | 237 | -37 | -49 | -42 | +8 | -31 | -47 |
Ezetimibe and Simvastatin Tablets 10/40Þ | 482 | -40¶ | -56¶ | -45¶ | +11¶ | -32 | -52¶ |
Week 24 | |||||||
Atorvastatin 80 mg | 228 | -40 | -53 | -45 | +6 | -35 | -50 |
Ezetimibe and Simvastatin Tablets 10/80Þ | 459 | -43¶ -59¶ | -49¶ | +12¶ | -35 | -55¶ | |
* For triglycerides, median % change from baseline. † Baseline -on no lipid-lowering drug. ‡ Atorvastatin: 10 mg start dose titrated to 20 mg, 40 mg, and 80 mg through Weeks 6, 12, 18, and 24. § Ezetimibe and simvastatin tablets: 10/10 start dose titrated to 10/20, 10/40, and 10/80 through Weeks 6, 12, 18, and 24. ¶ p≤0.05 for difference with atorvastatin in the specified week. # Ezetimibe and simvastatin tablets: 10/20 start dose titrated to 10/40, 10/40, and 10/80 through Weeks 6, 12, 18, and 24. Ϸ Data pooled for common doses of ezetimibe and simvastatin tablets at Weeks 18 and 24. | |||||||
In a multicenter, double-blind, 6-week study, 2959 patients with primary hyperlipidemia, who had not met their NCEP ATP III target LDL-C goal, were randomized to one of six treatment groups: Ezetimibe and simvastatin tablets (10/20, 10/40, or 10/80) or rosuvastatin (10 mg, 20 mg, or 40 mg).
The effects of ezetimibe and simvastatin tablets and rosuvastatin on total-C, LDL-C, Apo B, TG, non-HDL-C and HDL-C are shown in Table 11.
Table 11: Response to Ezetimibe and Simvastatin Tablets and Rosuvastatin in Patients with Primary Hyperlipidemia (Mean* % Change from Untreated Baseline†)
| Treatment (Daily Dose) | N | Total-C‡ | LDL-C‡ | Apo B‡ | HDL-C | TG* | Non-HDL-C‡ |
|---|---|---|---|---|---|---|---|
Ezetimibe and simvastatin tablets by dose | |||||||
10/20 | 476 | -37§ | -52§ | -42§ | +7 | -23§ | -47§ |
10/40 | 477 | -39¶ | -55¶ | -44¶ | +8 | -27 | -50¶ |
10/80 | 474 | -44# | -61# | -50# | +8 | -30# | -56# |
Rosuvastatin by dose | |||||||
10 mg | 475 | -32 | -46 | -37 | +7 | -20 | -42 |
20 mg | 478 | -37 | -52 | -43 | +8 | -26 | -48 |
40 mg | 475 | -41 | -57 | -47 | +8 | -28 | -52 |
* For triglycerides, median % change from baseline. † Baseline - on no lipid-lowering drug. ‡ Ezetimibe and simvastatin tablets doses pooled (10/20 to 10/80) provided significantly greater reductions in total-C, LDL-C, Apo B, and non-HDL-C compared to rosuvastatin doses pooled (10 to 40 mg). § p<0.05 vs. rosuvastatin 10 mg. ¶ p<0.05 vs. rosuvastatin 20 mg. # p<0.05 vs. rosuvastatin 40 mg. | |||||||
In a multicenter, double-blind, 24-week trial, 214 patients with type 2 diabetes mellitus treated with thiazolidinediones (rosiglitazone or pioglitazone) for a minimum of 3 months and simvastatin 20 mg for a minimum of 6 weeks were randomized to receive either simvastatin 40 mg or the coadministered active ingredients equivalent to ezetimibe and simvastatin tablets 10/20. The median LDL-C and HbA1c levels at baseline were 89 mg/dL and 7.1%, respectively.
Ezetimibe and simvastatin tablets 10/20 was significantly more effective than doubling the dose of simvastatin to 40 mg. The median percent changes from baseline for ezetimibe and simvastatin tablets vs. simvastatin were: LDL-C
-25% and -5%; total-C -16% and -5%; Apo B -19% and -5%; and non-HDL-C -23% and -5%. Results for HDL-C and TG between the two treatment groups were not significantly different.
Ezetimibe
In two multicenter, double-blind, placebo-controlled, 12-week studies in 1719 patients with primary hyperlipidemia, ezetimibe significantly lowered total-C (-13%), LDL-C (-19%), Apo B (-14%), and TG (-8%), and increased HDL-C (+3%) compared to placebo. Reduction in LDL-C was consistent across age, sex, and baseline LDL-C.
Simvastatin
In two large, placebo-controlled clinical trials, the Scandinavian Simvastatin Survival Study (N=4,444 patients) and the Heart Protection Study (N=20,536 patients), the effects of treatment with simvastatin were assessed in patients at high risk of coronary events because of existing coronary heart disease, diabetes, peripheral vessel disease, history of stroke or other cerebrovascular disease. Simvastatin was proven to reduce: the risk of total mortality by reducing CHD deaths; the risk of non-fatal myocardial infarction and stroke; and the need for coronary and non-coronary revascularization procedures.
No incremental benefit of ezetimibe and simvastatin tablets on cardiovascular morbidity and mortality over and above that demonstrated for simvastatin has been established.
14.2 Homozygous Familial Hypercholesterolemia (Hofh)
14.3 Chronic Kidney Disease (Ckd)
The Study of Heart and Renal Protection (SHARP) was a multinational, randomized, placebo-controlled, double-blind trial that investigated the effect of ezetimibe and simvastatin tablets on the time to a first major vascular event (MVE) among 9438 patients with moderate to severe chronic kidney disease (approximately one-third on dialysis at baseline) who did not have a history of myocardial infarction or coronary revascularization. An MVE was defined as nonfatal MI, cardiac death, stroke, or any revascularization procedure. Patients were allocated to treatment using a method that took into account the distribution of 8 important baseline characteristics of patients already enrolled and minimized the imbalance of those characteristics across the groups.
For the first year, 9438 patients were allocated 4:4:1, to ezetimibe and simvastatin tablets 10/20, placebo, or simvastatin 20 mg daily, respectively. The 1-year simvastatin arm enabled the comparison of ezetimibe and simvastatin tablets to simvastatin with regard to safety and effect on lipid levels. At 1 year the simvastatin-only arm was re-allocated 1:1 to ezetimibe and simvastatin tablets 10/20 or placebo. A total of 9270 patients were ever allocated to ezetimibe and simvastatin tablets 10/20 (n=4650) or placebo (n=4620) during the trial. The median follow-up duration was 4.9 years. Patients had a mean age of 61 years; 63% were male, 72% were Caucasian, and 23% were diabetic; and, for those not on dialysis at baseline, the median serum creatinine was 2.5 mg/dL and the median estimated glomerular filtration rate (eGFR) was 25.6 mL/min/1.73 m2, with 94% of patients having an eGFR < 45 mL/min/1.73 m2. Eligibility did not depend on lipid levels. Mean LDL-C at baseline was 108 mg/dL. At 1 year, the mean LDL-C was 26% lower in the simvastatin arm and 38% lower in the ezetimibe and simvastatin tablets arm relative to placebo. At the midpoint of the study (2.5 years), the mean LDL-C was 32% lower for ezetimibe and simvastatin tablets relative to placebo. Patients no longer taking study medication were included in all lipid measurements.
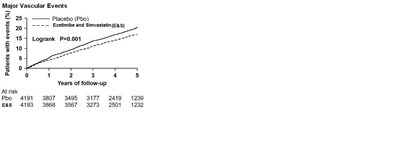
In the primary intent-to-treat analysis, 639 (15.2%) of 4193 patients initially allocated to ezetimibe and simvastatin tablets and 749 (17.9%) of 4191 patients initially allocated to placebo experienced an MVE. This corresponded to a relative risk reduction of 16% (p=0.001) (see Figure 1). Similarly, 526 (11.3%) of 4650 patients ever allocated to ezetimibe and simvastatin tablets and 619 (13.4%) of 4620 patients ever allocated to placebo experienced a major atherosclerotic event (MAE; a subset of the MVE composite that excluded non-coronary cardiac deaths and hemorrhagic stroke), corresponding to a relative risk reduction of 17% (p=0.002). The trial demonstrated that treatment with ezetimibe and simvastatin tablets 10/20 mg versus placebo reduced the risk for MVE and MAE in this CKD population. The study design precluded drawing conclusions regarding the independent contribution of either ezetimibe or simvastatin to the observed effect.
The treatment effect of ezetimibe and simvastatin tablets on MVE was attenuated among patients on dialysis at baseline compared with those not on dialysis at baseline. Among 3023 patients on dialysis at baseline, ezetimibe and simvastatin tablets reduced the risk of MVE by 6% (RR 0.94: 95% CI 0.80 to 1.09) compared with 22% (RR 0.78: 95% CI 0.69 to 0.89) among 6247 patients not on dialysis at baseline (interaction P=0.08).
Figure 1: Effect of Ezetimibe and Simvastatin Tablets on the Primary Endpoint of Risk of Major Vascular Events |
The individual components of MVE in all patients ever allocated to ezetimibe and simvastatin tablets or placebo are presented in Table 12.
Table 12: Number of First Events for Each Component of the Major Vascular Event Composite Endpoint in SHARP*
Outcome | Ezetimibe and Simvastatin Tablets 10/20 (N=4650) | Placebo (N=4620) | Risk Ratio (95% CI) | P-value |
Major Vascular Events | 701 (15.1%) | 814 (17.6%) | 0.85 (0.77 to 0.94) | 0.001 |
Nonfatal MI | 134 (2.9%) | 159 (3.4%) | 0.84 (0.66 to 1.05) | 0.12 |
Cardiac Death | 253 (5.4%) | 272 (5.9%) | 0.93 (0.78 to 1.10) | 0.38 |
Any Stroke | 171 (3.7%) | 210 (4.5%) | 0.81 (0.66 to 0.99) | 0.038 |
| 131 (2.8%) | 174 (3.8%) | 0.75 (0.60 to 0.94) | 0.011 |
| 45 (1%) | 37 (0.8%) | 1.21 (0.78 to 1.86) | 0.4 |
Any Revascularization | 284 (6.1%) | 352 (7.6%) | 0.79 (0.68 to 0.93) | 0.004 |
*Intention-to-treat analysis on all SHARP patients ever allocated to ezetimibe and simvastatin tablets or placebo. | ||||
Among patients not on dialysis at baseline, ezetimibe and simvastatin tablets did not reduce the risk of progressing to end-stage renal disease compared with placebo (RR 0.97: 95% CI 0.89 to 1.05).
16 How Supplied/Storage And Handling
Storage
17 Patient Counseling Information
17.1 Muscle Pain
17.2 Liver Enzymes
17.3 Pregnancy
17.4 Breastfeeding
*Trademarks are the properties of their respective owners.
Other
Manufactured for:
Northstar Rx LLC
Memphis, TN 38141.
Manufactured by:
Glenmark Pharmaceuticals Ltd.
Pithampur, Madhya Pradesh 454775, India
November 2020
Spl Patient Package Insert
Patient Information
Ezetimibe and Simvastatin
(e zet' i mibe and sim" va stat' in)
Tablets
Read this Patient Information carefully before you start taking ezetimibe and simvastatin tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about ezetimibe and simvastatin tablets, ask your doctor. Only your doctor can determine if ezetimibe and simvastatin tablets are right for you.
What are ezetimibe and simvastatin tablets?
Ezetimibe and simvastatin tablets are a prescription medicine that contains 2 cholesterol lowering medicines, ezetimibe and simvastatin. Ezetimibe and simvastatin tablets are used along with diet to:
• lower the level of your "bad" cholesterol (LDL)• increase the level of your "good" cholesterol (HDL)• lower the level of fat in your blood (triglycerides)• have been taking ezetimibe and simvastatin tablets 10/80 mg chronically (such as 12 months or more) without having muscle damage.• do not need to take certain other medicines with ezetimibe and simvastatin tablets that would increase your chance of getting muscle damage.• Certain anti-fungal medicines including:• itraconazole• ketoconazole• posaconazole• voriconazole
• HIV protease inhibitors (indinavir, nelfinavir, ritonavir, saquinavir, tipranavir, or atazanavir)• Certain hepatitis C virus protease inhibitors (such as boceprevir or telaprevir)• Certain antibiotics, including:• erythromycin• clarithromycin• telithromycin
• nefazodone• medicines containing cobicistat• A fibric acid medicine for lowering cholesterol called gemfibrozil• cyclosporine• danazol• are allergic to ezetimibe or simvastatin or any of the ingredients in ezetimibe and simvastatin tablets. See the end of this leaflet for a complete list of ingredients in ezetimibe and simvastatin tablets.• have liver problems.• are pregnant or plan to become pregnant. Ezetimibe and simvastatin tablets may harm your unborn baby. If you are a woman of childbearing age, you should use an effective method of birth control to prevent pregnancy while using ezetimibe and simvastatin tablets. If you become pregnant while taking ezetimibe and simvastatin tablets, stop taking ezetimibe and simvastatin tablets and call your doctor.• are breastfeeding or plan to breastfeed. It is not known if ezetimibe and simvastatin passes into your breast milk. You and your doctor should decide the best way to feed your baby if you take ezetimibe and simvastatin tablets.• have unexplained muscle aches or weakness• have kidney problems• have or have had liver problems or drink more than 2 glasses of alcohol daily• have thyroid problems• are 65 years of age or older• are Chinese• fibric acid derivatives (such as fenofibrate)• amiodarone or dronedarone (drugs used to treat an irregular heartbeat)• verapamil, diltiazem, amlodipine, or ranolazine (drugs used to treat high blood pressure, chest pain associated with heart disease, or other heart conditions)• grapefruit juice (which should be avoided while taking ezetimibe and simvastatin tablets)• colchicine (a medicine used to treat gout)• lomitapide (a medicine used to treat a serious and rare genetic cholesterol condition)• daptomycin (a drug used to treat complicated skin and bloodstream infections)• large doses of niacin or nicotinic acid• Take ezetimibe and simvastatin tablets exactly as your doctor tells you to take it.• Do not change your dose or stop taking ezetimibe and simvastatin tablets without talking to your doctor.• Take ezetimibe and simvastatin tablets 1 time each day in the evening.• Take ezetimibe and simvastatin tablets with or without food.• While taking ezetimibe and simvastatin tablets, continue to follow your cholesterol-lowering diet and to exercise as your doctor told you to.• If you miss a dose, do not take an extra dose. Just resume your usual schedule.• Your doctor should do fasting blood tests to check your cholesterol while you take ezetimibe and simvastatin tablets. Your doctor may change your dose of ezetimibe and simvastatin tablets if needed.• If you take too much ezetimibe and simvastatin tablets, call your doctor or Poison Control Center at 1-800-222-1222 or go to the nearest hospital emergency room right away.• Muscle pain, tenderness and weakness (myopathy). Muscle problems, including muscle breakdown, can be serious in some people and rarely cause kidney damage that can lead to death.• you have unexplained muscle pain, tenderness, or weakness, especially if you have a fever or feel more tired than usual, while you take ezetimibe and simvastatin tablets.• you have muscle problems that do not go away even after your doctor has advised you to stop taking ezetimibe and simvastatin tablets. Your doctor may do further tests to diagnose the cause of your muscle problems.• are taking certain other medicines while you take ezetimibe and simvastatin tablets• are 65 years of age or older• are female• have thyroid problems (hypothyroidism) that are not controlled• have kidney problems• are taking higher doses of ezetimibe and simvastatin tablets, particularly the 10/80 mg dose• are Chinese• Liver problems. Your doctor should do blood tests to check your liver before you start taking ezetimibe and simvastatin tablets and if you have any symptoms of liver problems while you take ezetimibe and simvastatin tablets. Call your doctor right away if you have the following symptoms of liver problems:• loss of appetite• upper belly pain• dark urine• yellowing of your skin or the whites of your eyes• feel tired or weak
• headache• increased liver enzyme levels• muscle pain• upper respiratory infection• diarrhea• allergic reactions including swelling of the face, lips, tongue, and/or throat that may cause difficulty in breathing or swallowing (which may require treatment right away), rash, hives; joint pain; inflammation of the pancreas; nausea; dizziness; tingling sensation; depression; gallstones; trouble sleeping; poor memory; memory loss; confusion; erectile dysfunction; breathing problems including persistent cough and/or shortness of breath or fever.• Store ezetimibe and simvastatin tablets at room temperature between 20°C to 25°C (68°F to 77°F).• Keep ezetimibe and simvastatin tablets in its original container until you use it.• Keep ezetimibe and simvastatin tablets in a tightly closed container, and keep ezetimibe and simvastatin tablets out of light.
Ezetimibe and simvastatin tablets are for patients who cannot control their cholesterol levels by diet and exercise alone.
Ezetimibe and simvastatin tablets have not been shown to reduce heart attacks or strokes more than simvastatin alone.
It is not known if ezetimibe and simvastatin tablets are safe and effective in children under 10 years of age or in girls who have not started their period (menses).
The usual dose of ezetimibe and simvastatin tablets is 10/10 mg to 10/40 mg 1 time each day.
Ezetimibe and simvastatin tablets 10/80 mg increases your chance of developing muscle damage. The 10/80 mg dose should only be used by people who:
If you are unable to reach your LDL-cholesterol goal using ezetimibe and simvastatin tablets 10/40 mg, your doctor should switch you to another cholesterol-lowering medicine.
Who should not take ezetimibe and simvastatin tablets?
Do not take ezetimibe and simvastatin tablets if you take:
Ask your doctor or pharmacist for a list of these medicines if you are not sure.
Also do not take ezetimibe and simvastatin tablets if you:
What should I tell my doctor before and while taking ezetimibe and simvastatin tablets?
Tell your doctor if you:
Also see "What are the possible side effects of ezetimibe and simvastatin tablets?"
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Tell your doctor who prescribes ezetimibe and simvastatin tablets if another doctor increases the dose of another medicine you are taking.
Talk to your doctor before you start taking any new medicines.
Taking ezetimibe and simvastatin tablets with certain other medicines may affect each other causing side effects. Ezetimibe and simvastatin tablets may affect the way other medicines work, and other medicines may affect how ezetimibe and simvastatin tablets works.
Taking ezetimibe and simvastatin tablets with certain substances can increase the risk of muscle problems. It is especially important to tell your doctor if you take:
Tell your doctor if you are taking niacin or a niacin-containing product, as this may increase your risk of muscle problems, especially if you are Chinese.
It is also important to tell your doctor if you are taking coumarin anticoagulants (drugs that prevent blood clots, such as warfarin).
Tell your doctor about all the medicines you take, including any prescription and nonprescription medicines, vitamins, and herbal supplements.
How should I take ezetimibe and simvastatin tablets?
What are the possible side effects of ezetimibe and simvastatin tablets?
Ezetimibe and simvastatin tablets may cause serious side effects, including:
The most common side effects of ezetimibe and simvastatin tablets include:
Additional side effects that have been reported in general use with ezetimibe and simvastatin tablets or with ezetimibe or simvastatin tablets (tablets that contain the active ingredients of ezetimibe and simvastatin tablets) include:
Tell your doctor if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of ezetimibe and simvastatin tablets. For more information, ask your doctor or pharmacist.
Call your doctor about medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ezetimibe and simvastatin tablets?
Keep ezetimibe and simvastatin tablets and all medicines out of the reach of children.
General Information about the safe and effective use of ezetimibe and simvastatin tablets.
Ezetimibe and simvastatin tablets work to reduce your cholesterol in two ways. It reduces the cholesterol absorbed in your digestive tract, as well as the cholesterol your body makes by itself. Ezetimibe and simvastatin tablets do not help you lose weight.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ezetimibe and simvastatin tablets for a condition for which it was not prescribed. Do not give ezetimibe and simvastatin tablets to other people, even if they have the same condition that you have. It may harm them.
This Patient Information summarizes the most important information about ezetimibe and simvastatin tablets. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about ezetimibe and simvastatin tablets that is written for health professionals.
For more information, please call 1-800-206-7821.
What are the ingredients in ezetimibe and simvastatin tablets?
Active Ingredients: ezetimibe and simvastatin
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Northstar Rx LLC
Memphis, TN 38141.
Manufactured by:
Glenmark Pharmaceuticals Ltd.
Pithampur, Madhya Pradesh 454775, India
November 2020
Principal Display Panel - 10 Mg/10 Mg Tablet Bottle Label
NDC 16714-778-01
Ezetimibe and Simvastatin Tablets
10 mg/10 mg
Principal Display Panel - 10 Mg/20 Mg Tablet Bottle Label
NDC 16714-779-01
Ezetimibe and Simvastatin Tablets
10 mg/20 mg
Principal Display Panel - 10 Mg/40 Mg Tablet Bottle Label
NDC 16714-780-01
Ezetimibe and Simvastatin Tablets
10 mg/40 mg
Principal Display Panel - 10 Mg/80 Mg Tablet Bottle Label
NDC 16714-781-01
Ezetimibe and Simvastatin Tablets
10 mg/80 mg
* Please review the disclaimer below.