Product Images Simvastatin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Simvastatin NDC 16729-005 by Accord Healthcare, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 mg- 30 tab - simvastatin 10mg 30

Each tablet contains Simvastatin USP 10 mg. It is advised to store the tablets at a temperature range of 20°C to 25°C (68°F to 77°F). These tablets are manufactured and distributed by Accord Healthcare, Inc. and Intas Pharmaceuticals Limited respectively. The package contains 30 tablets. Additional information, including warnings, may be present but is not available due to the quality of the text.*
20 mg- 30 tab - simvastatin 20mg 30

Each tablet of this medicine contains an unknown ingredient with code number NOC 16726-005-10. The recommended storage temperature is between 20-25°C. The manufacturer of this medicine is Accord Healthcares, located in Durham, with another manufacturing unit in Hmacabad, India. The rest of the text seems to be gibberish or unreadable.*
40 mg- 30 tab - simvastatin 40mg 30

Each tablet contains Simvastatin USP 40 mg. This medication is usually prescribed for adult patients to manage cholesterol levels. The temperature for storage should be between 20°C to 25°C (68°F to 77°F). The tablets are USP controlled and manufactured for Accord Healthcare in Durham. The manufacturer of the tablets is Intas Pharmaceuticals Limited based in India. The rest of the text is not readable.*
5 mg- 30 tab - simvastatin 5mg 30

Each tablet contains 5mg of Simvastatin USP. The usual adult dosage is not available. Reference is made to a package insert for dosage information. The tablet should be stored at controlled room temperature. The tablet is manufactured for Accord Healthcare, Inc. in India by Intas Pharmaceuticals Limited. There is a varnish free area on the tablet measuring 12x36mm.*
80 mg- 30 tab - simvastatin 80mg 30
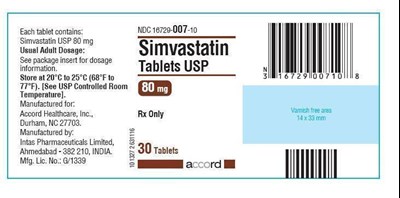
Each tablet of Simvastatin USP contains 80mg of the active ingredient. It is recommended to refer to the package insert for dosage information. The tablets should be stored between 20°C to 25°C (68°F to 77°F). The manufacturer for Accord Healthcare, Inc. is located in Durham, NC. The tablets are manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India. The package contains 30 tablets.*
Simvastatin - simvastatin fig1

This is a table presenting the baseline characteristics of 11 patients in a study, divided into subgroups based on the presence or absence of various medical conditions. The conditions include diabetes mellitus and peripheral vascular disease. There are also subgroups based on gender, age, and cholesterol levels. The table shows the incidence of major vascular events and major coronary events in these subgroups, represented as relative risk. The study involves the use of simvastatin tablets, and there is a comparison with a placebo group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
