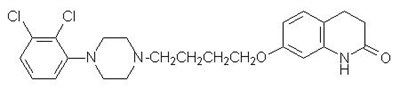Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
- 10 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 10mg 100 tab carton
- 10 mg-30 Tablets - aripiprazole 10mg 30 tab
- 15 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 15mg 100 tab carton
- 15 mg-30 Tablets - aripiprazole 15mg 30 tab
- 20 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 20mg 100 tab carton
- 20 mg-30 Tablets - aripiprazole 20mg 30 tab
- 2 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 2mg 100 tab carton
- 2 mg-30 Tablets - aripiprazole 2mg 30 tab
- 30 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 30mg 100 tab carton
- 30 mg-30 Tablets - aripiprazole 30mg 30 tab
- 5 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 5mg 100 tab carton
- 5 mg-30 Tablets - aripiprazole 5mg 30 tab
- Figure 1 - aripiprazole figure 1
- Figure 2 - aripiprazole figure 2
- Figure 3 - aripiprazole figure 3
- Figure 4 - aripiprazole figure 4
- Figure 5 - aripiprazole figure 5
- Figure-6 - aripiprazole figure 6
- Figure-7 - aripiprazole figure 7
- Figure-8 - aripiprazole figure 8
- aripiprazole chemical structure - aripiprazole structure
Product Label Images
The following 21 images provide visual information about the product associated with Aripiprazole NDC 16729-282 by Accord Healthcare, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 10mg 100 tab carton

This appears to be the label information for a medication called Aripiprazole. It is available in tablet form and comes in a pack of 100 unit-dose tablets with each tablet containing 10mg of the medication. The label specifies that the medication is only available with a prescription and includes a guidance document for dispensing the medication.*
15 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 15mg 100 tab carton

This is a description of a medication called Aripiprazole which comes in the form of tablets. The text includes the dosage amount (15mg) and the number of tablets in each container (100). There are instructions for pharmacists to dispense a medication guide to each patient, and the medication is only to be taken with a prescription (Rx Only). There are also codes, numbers, and symbols related to the medication that are not fully understandable from the text.*
15 mg-30 Tablets - aripiprazole 15mg 30 tab

Each uncoated tablet contains Aripiprazole USP 15 mg. This medication is manufactured by Accord Healthcares, Inc. It should be stored in tightly closed containers at 25°C (T7°F). The excursions between 157G to 30°C (39°Fto 86°F) are permitted. The medication guide is available at https://accordhealthcare.us/medication-guides/. The accompanying package insert contains indications and dosage information. The tablets are manufactured by Intas Pharmaceuticals Limited, Pharmez, Ahmedabad-382.213, INDIA. Rx Only.*
20 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 20mg 100 tab carton

This is a medication called Aripiprazole which comes in the form of tablets. The medication guide should be given to each patient by the pharmacist. The medication is available in a pack of 100-tablets with each tablet being unit-dose.*
20 mg-30 Tablets - aripiprazole 20mg 30 tab

Each uncoated tablet contains Aripiprazole USP 20 mg. The product comes with an accompanying package insert that provides indications and dosage information. It should be stored in tightly closed containers at a temperature range between 15°C to 30°C (59°F to 86°F). The medication guide is available at accordhealthcare.us/medication-guides. The tablets are manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India, and marketed by Accord Healthcare Inc. This product is available by prescription only. The pharmacist must dispense the enclosed medication guide to each patient. The package contains 30 tablets. The lot and expiry date information is to be overcoded on the blank area with a varnish-free label of dimensions 45X22mm.*
2 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 2mg 100 tab carton

This is a medication package of Aripiprazole Tablets, USP with a strength of 16729-278.46. Each pack contains 100 unit-dose tablets. The pharmacist is advised to dispense the medication guide enclosed with each patient.*
2 mg-30 Tablets - aripiprazole 2mg 30 tab

Each uncoated tablet contains 2mg of Aripiprazole USP. The medication is stored in tightly closed containers at a temperature between 15°C to 30°C. The medication guide is available at accordhealthcare.us/medication-guides. The tablets are manufactured by Intas Pharmaceuticals Limited in India and distributed by Accord Healthcare, Inc. Each package contains 30 tablets and includes dosage information and indications. The medication guide included in the package should be dispensed to each patient by the pharmacist. The Lot and EXP information is meant to be overcoded in the blank area. NDC code for the product is 16729-278-10.*
30 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 30mg 100 tab carton

The text is a list of medication information for Aripiprazole Tablets, USP, including dosage and packaging details. It also includes a message to pharmacists to dispense a medication guide to each patient.*
30 mg-30 Tablets - aripiprazole 30mg 30 tab

Each uncoated tablet contains 30 mg of Aripiprazole USP, which is a prescription medication. Store it in tightly closed containers at a temperature between 15-30°C. Medication guides are available online at the provided link. The manufacturer's details, including their address and license number, are listed. It is important to dispense the enclosed medication guide to each patient. This product comes in a blister pack of 30 tablets. Overall, this is a medication used in the treatment of certain mental disorders.*
5 mg-100 (10 x 10) Unit-dose Tablets - aripiprazole 5mg 100 tab carton

This is a description for Aripiprazole Tablets, USP, which comes in a pack of 100 (10 x 10) Unit-dose Tablets and is RxOnly. It is indicated for treating mental / mood disorders like bipolar disorder, schizophrenia, depression, and irritability associated with autistic disorder. It is important for pharmacists to provide the enclosed medication guide to each patient.*
5 mg-30 Tablets - aripiprazole 5mg 30 tab

Each uncoated tablet contains 5 mg of Arpiprazole USP. There is an accompanying package insert for indications and dosage information. These tablets should be stored in tightly closed containers at 25°C (T7°F), with excursions permitted between 157G to 30°G (39°F to 86°F). Medication guides are available at https://www.accordhealthcare.us/medication-guides. This product is manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India, and distributed by Accord Healthcare, Inc. The lot number and expiration date information should not be added to the blank area, which must remain varnish-free for overcoding purposes. This is a prescription medication. Pharmacists should dispense the enclosed medication guide to each patient. Not available for non-English characters.*
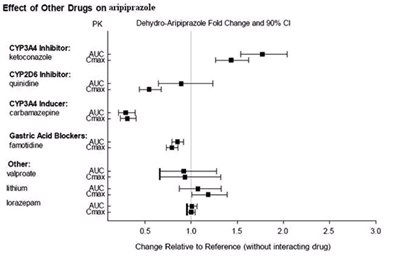
Figure 1 - aripiprazole figure 1

The text describes the effect of various drugs on aripiprazole, including inhibitors and inducers of CYP enzymes, gastric acid blockers, and other drugs. The table includes PK (pharmacokinetic) parameters such as Aipprazol Fad change and % CI (confidence interval) for each drug. The impact of the drugs on aripiprazole can be seen in the change relative to the reference (without interacting drug).*
Figure 3 - aripiprazole figure 3

This appears to be a table showing the effect of aripiprazole on various drugs and their metabolic pathways. The level of change in percentage is shown for each drug, as well as the metabolic pathway it interacts with. It is not clear what the exact units or values represent, but the table appears to be a reference for healthcare professionals.*
Figure 4 - aripiprazole figure 4

This is a report on different special populations focusing on gender and liver or renal impairments. The report also includes charts displaying the average AUC (area under the curve) of these populations, specifically comparing female versus male individuals and those with liver or renal impairments versus those without. Additionally, there is a chart depicting aviprazcie foci change and 50% CI.*
Figure-8 - aripiprazole figure 8

This is a table showing the proportion of subjects at risk for relapse on ARFPRAZOLE. The drug's efficacy is likely being tested, with ARPIBRAZOLE being the experimental group and PucEsq being the control group. There are numerical values under the two groups, but their meaning is difficult to ascertain.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.