Product Images Trazodone Hydrochloride
View Photos of Packaging, Labels & Appearance
- 100mg-100bt blister - trazo 100mg 100bt blister
- 100mg-100bt carton - trazo 100mg 100bt carton
- 100mg-100btl - trazo 100mg 100bt label
- 100mg-break - trazo 100mg break
- 100mg-score - trazo 100mg score
- 150mg-100btl - trazo 150mg 100btl
- 150mg-break - trazo 150mg break
- 150mg-tablet - trazo 150mg tablet
- 200mg-break - trazo 200mg break
- 300mg-100btl - trazo 300mg 100btl
- 300mg-break - trazo 300mg break
- 50mg-100bt blister - trazo 50mg 100bt blister
- 50mg-100bt carton - trazo 50mg 100bt carton
- 50mg-100bt label - trazo 50mg 100bt label
- 50mg-score - trazo 50mg score
- 75mg-score - trazo 75mg score
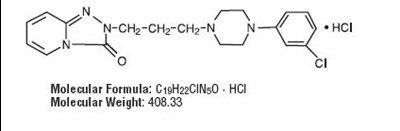
- structure - trazo structure
Product Label Images
The following 17 images provide visual information about the product associated with Trazodone Hydrochloride NDC 16729-299 by Accord Healthcare, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100mg-100bt carton - trazo 100mg 100bt carton

This is a medication label for Trazodone Hydrochloride Tablets, used to treat depression. It includes information such as the drug dosage, manufacturer details, and storage instructions. The label also informs the pharmacist to dispense a medication guide to each patient, and suggests using child-resistant containers if this medication is dispensed for outpatient use.*
100mg-100btl - trazo 100mg 100bt label

Each tablet contains trazodone hydrochloride and comes in a light-resistant container. The usual dosage information can be found in the package insert. Manufacturer information is also included.*
300mg-100btl - trazo 300mg 100btl

Each tablet contains 300mg of Trazodone Hydrochloride USP. The pharmacist is advised to instruct patients on the correct way to use the tablet, as it is designed to be broken. The medication guide is available at a specific website. The tablets should be stored in a tight, light-resistant container at temperatures ranging from 68°F to 77°F. Developed by Accord Healthcars, Inc. in Durham, NC 27703, and manufactured in Pharmez, Ahmedabad-382 213, INDIA, these tablets are sold in packages of 100. Information about licensing, lot numbers, and expiration dates is provided.*
50mg-100bt carton - trazo 50mg 100bt carton

This text provides information regarding Trazodone Hydrochloride Tablets, USP. It suggests that 100 unit-dose tablets containing 50mg of Trazodone Hydrochloride USP are packed in the enclosed medication guide, which should be dispensed to each patient. The tablets should be stored within a certain temperature range. A child-resistant container should be used for outpatient use. The manufacturer is Accord Healthcare, Inc., while the product is distributed by Intas Pharmaceuticals Limited in India. A pharmacist should administer the medication on prescription only.*
50mg-100bt label - trazo 50mg 100bt label

This text fragment seems to be a mix of product descriptions and instructions, including recommended storage conditions, dosage instructions, and medication guide availability. However, it is not clear what kind of product is being marketed.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.