Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 15 images provide visual information about the product associated with Tadalafil NDC 16729-370 by Accord Healthcare Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Tadalafil Tablets, USP 2.5 mg - Carton - tadalafil carton 25mg
This text contains information about a medication named Tadalafil, available in tablet form for once-daily use. The medication comes in a box that contains 2 strips of 15 tablets, with each strip labeled with the days of the week. Patients should begin taking a tablet labeled with the day of the week they start and continue taking one tablet every day at the same time, without regard to meals. The tablet in the center of the strip is the fastest tablet and should be taken after the 30th tablet. This medication is only available with a prescription, and it should be kept out of reach of children. There is also mention of an unvarnished area for overcoding and a varnish-free area for passing, but it is unclear what those mean in this context.*
Tadalafil Tablets, USP 5 mg - Carton - tadalafil carton 5mg
This is a description of a medication called Tadalafil in a tablet form. The medication is meant for once-daily use and comes in a box containing two blister strips with 15 tablets in each strip. Each strip is labeled with the days of the week, and the patient should start with the tablet labeled with the day they begin taking the medication. The tablets should be taken at the same time every day, regardless of meals. The tablet in the center of the strip is the last tablet in the strip, and after the 30th tablet, the patient may begin another box. The warning on the package advises to keep out of reach of children. The dosage instructions advise to read the accompanying instructions before taking the medication.*
Figure - tadalafil figure1
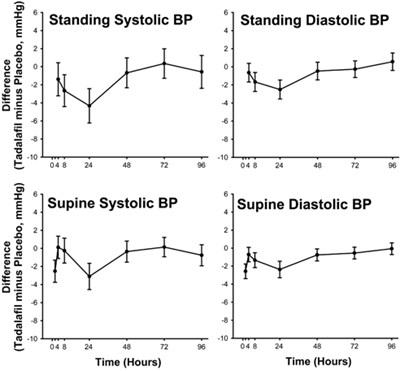
The text appears to be a table containing blood pressure measurements for patients taking Tadalafil and Placebo medications. The table shows the difference in blood pressure (in mmHg) between Tadalafil and Placebo for standing systolic and diastolic pressures, as well as supine systolic and diastolic pressures, over a period of time in hours.*
Figure - tadalafil figure2
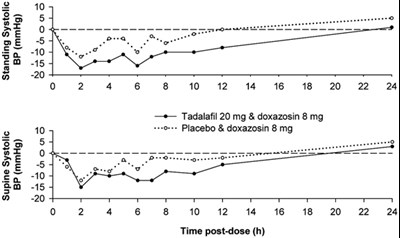
The text seems to be a mix of numerical, currency, and letter/word characters with a chart showing the time post-dose. It may be related to a medical study involving the comparison of Tadalafil and doxazosin, as well as a placebo and doxazosin. However, without further context, it is not possible to provide a definitive description.*
Figure - tadalafil figure4
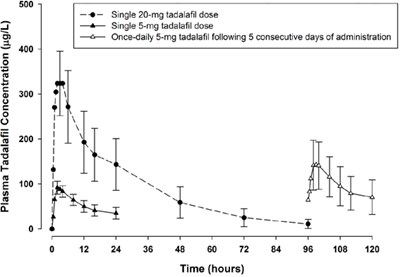
This text represents a chart displaying plasma concentration levels of Tadalafil, a medication used in the treatment of erectile dysfunction. The chart shows the concentration levels measured in ug/L at various time intervals, following administration of single 20mg and 5mg doses of Tadalafil, as well as once-daily 5mg doses for five consecutive days. The time intervals are represented on the X-axis in hours, and the concentration levels on the Y-axis. No further information is available.*
Figure 5 - tadalafil figure5
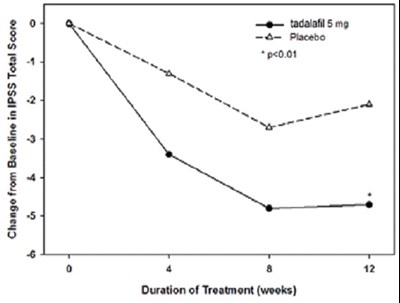
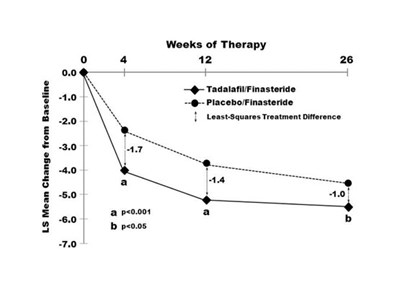
The text describes the change in the IPSS Total Score for tadalafil and placebo treatment over a period of 12 weeks, with a significant difference (p<0.01) observed between the two groups. No further information is provided.*
Tadalafil Tablets, USP 20 mg - Label - tadalafil label 20mg
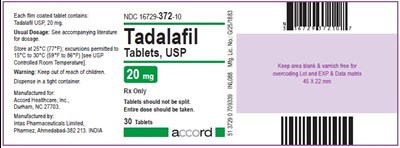
This is a description of Tadalafil USP 20mg, which is contained in coated tablets. The NDC number for the tablets is 16728-372-10. The usual dosage instructions are included in the accompanying literature. The tablets should not be split, and the entire dose should be taken. The manufacturer is Intas Pharmaceuticals Limited, and the tablets are dispensed in a container measuring 45x22mm. The text includes some instructions regarding storage and warns against overcosing, keeping the area free of varnish. The lot and expiration dates are also provided.*
Tadalafil Tablets, USP 2.5 mg - Label - tadalafil label 25mg
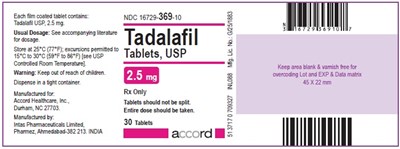
This is a description of a medication called Tadalafil Tablets, USP which come in a bottle containing 30 tablets of 25 mg each. The recommended dosage is not given but is available in accompanying literature. It should be stored at a temperature between 15°C and 30°C. The medication should be kept away from the reach of children. The manufacturer is Accord Healthcare and it is distributed by Inta Pharmaceuticals Limited in India. There is a warning not to split the tablets and to take the entire dose. The text contains a lot number and expiration date but does not include instructions for use or possible side effects.*
Tadalafil Tablets, USP 5 mg - Label - tadalafil label 5mg
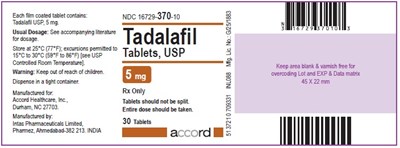
This appears to be information related to a medication called Tadalafil. The text mentions the NDC code for the medication, its dosage, and the fact that it comes in tablet form. There is also information about the manufacturer, Accord Healthcare, Inc., and instructions not to split the tablets and to take the entire dose. There does not appear to be any accompanying literature for the medication dosage.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.