Product Images Fulvestrant
View Photos of Packaging, Labels & Appearance
- Fulvestrant - fulvestrant injection carton
- chemical structure Fulvestrant - fulvestrant injection chemical structure
- Figure 1 - fulvestrant injection figure 1
- figure_10 - fulvestrant injection figure 10
- Figure 11 - fulvestrant injection figure 11
- Figure 12 - fulvestrant injection figure 12
- Figure 13 - fulvestrant injection figure 13
- Figure 14 - fulvestrant injection figure 14
- Figure 2 - fulvestrant injection figure 2
- Figure 3 - fulvestrant injection figure 3
- Figure 4 - fulvestrant injection figure 4
- Figure 5 - fulvestrant injection figure 5
- fig 6 kaplan-maier PFS: Study 1 ITT Population - fulvestrant injection figure 6
- Figure 7 Kaplan-Meier OS (Minimum Follow-up Duration of 50 Months): CONFIRM ITT Population - fulvestrant injection figure 7
- Figure_8.jpg - fulvestrant injection figure 8
- figure_9 - fulvestrant injection figure 9
- Fulvestrant - fulvestrant injection label
Product Label Images
The following 17 images provide visual information about the product associated with Fulvestrant NDC 16729-436 by Accord Healthcare Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Fulvestrant - fulvestrant injection carton

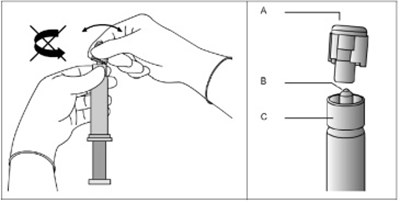
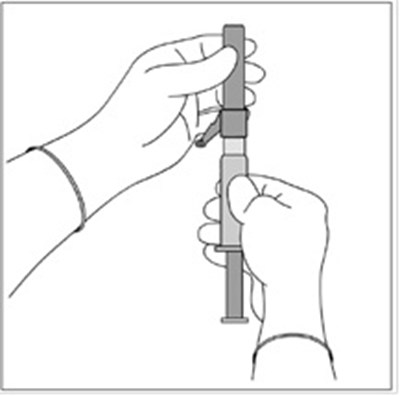
This text seems to be a combination of medical instructions and product information for Fulvestrant injection, a medication used for treating breast cancer. It includes advice on proper handling, storing, administration procedures, and dosages. It also highlights the presence of two SafetyGlide™ shielding intramuscular injection needles and mentions the need for their proper discard after use.*
Figure 12 - fulvestrant injection figure 12

This appears to be a graph showing the results of a study comparing the effectiveness of two drugs, Fulvestrant plus Abemaciclib and Fulvestrant plus Placebo, on Palionks at ok Time (months). There were 446 participants in the Fulvestrant plus Abemaciclib group and 223 in the Fulvestrant plus Placebo group. The graph shows that the Fulvestrant plus Abemaciclib group had higher Palionks scores compared to the Fulvestrant plus Placebo group. There were 100 censored observations, and the time range was from 0 to 57 months.*
fig 6 kaplan-maier PFS: Study 1 ITT Population - fulvestrant injection figure 6
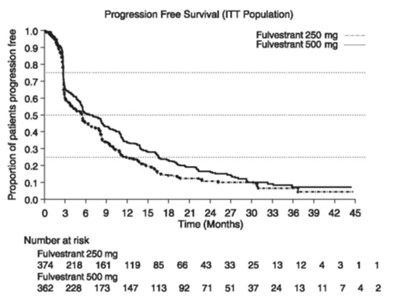
This appears to be a table displaying results from a study or clinical trial involving two different dosages of the drug Fulvestrant (250mg and 500mg). The numbers listed under "Number at risk" likely show how many participants were still being monitored at different time intervals (measured in months) throughout the study. The numbers in the subsequent columns likely show how many of those participants experienced some outcome or event of interest. Without additional context or information about what the study was measuring, it is difficult to provide a more detailed description.*
Figure 7 Kaplan-Meier OS (Minimum Follow-up Duration of 50 Months): CONFIRM ITT Population - fulvestrant injection figure 7
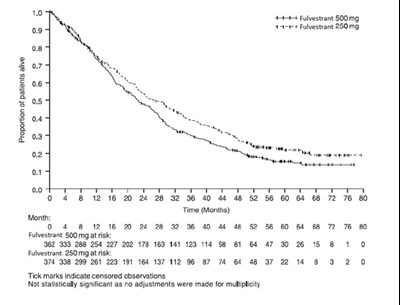
This is a graph showing the proportion of patients alive over time. Two treatments are compared: Fulvestrant 500mg and Fulvestrant 250mg. The X-axis shows the time in months, and the Y-axis shows the percentage of patients alive. The graph suggests that patients treated with Fulvestrant 500mg had a higher proportion of survival compared to those treated with 250mg, but the difference is not statistically significant. The graph also indicates that some patients’ observations were censored.*
Fulvestrant - fulvestrant injection label

This is a product description of a fulvestrant injection with a concentration of 50mg/mL, administered intramuscularly with a 500mg dose requiring two pre-filled syringes. The manufacturer is Intas Pharmaceuticals Limited and the product is distributed by IN5125. The NDC code is 16729-436-31.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.