Product Images Lorazepam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Lorazepam NDC 17478-040 by Akorn, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
lor06 0010 03
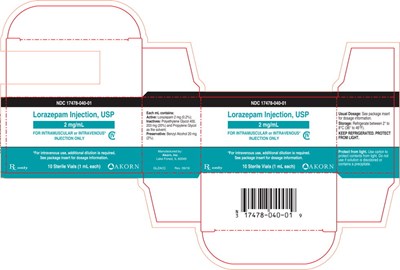
Lorazepam Injection, USP is a medication that is administered intravenously or intramuscularly and should be kept refrigerated. It is available in 1 mL vials, and additional dosage information can be found in the package insert. The usual dosage should be determined by a healthcare professional. The NDC codes for this medication are 17476-040-01 and 17478-040-01.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

